Background and Challenges
Quasi-solid-state lithium metal batteries (QSSLMBs), owing to their ultrahigh theoretical energy density exceeding 500 Wh kg⁻¹, have emerged as one of the most promising candidates for next-generation energy storage technologies. However, their practical application is severely hindered by critical interfacial challenges. The rigid nature of solid–solid contact confines the reactive region and leads to low active material utilization. Meanwhile, the drastic volume fluctuation of lithium metal anodes during cycling generates substantial mechanical stress, which induces interfacial delamination and increased impedance. In addition, the conventional planar contact geometry restricts ion transport and causes localized lithium plating/stripping, thereby aggravating dendrite growth and eventual cell failure. These issues become even more pronounced under high-loading and high-current-density conditions. Existing interfacial engineering strategies are generally unable to simultaneously achieve dynamic volume accommodation, dendrite suppression, and efficient charge transport, thus severely limiting the development of high-energy-density QSSLMBs.
Literature Introduction
Previous studies have mainly focused on improving interfacial wettability through coating modification or polymer interlayers, yet these approaches fail to accommodate the dynamic volume change during long-term cycling. Although integrated architectural designs can provide seamless contact, they still suffer from poor mechanical robustness and insufficient lithiophilicity, and thus cannot effectively address stress management during lithium deposition. In this work, an innovative gel electrolyte–anode integrated strategy based on an ion/electron mixed-conducting network is proposed. Through in situ polymerization, the gel electrolyte (PDOL) is infiltrated into a three-dimensional hollow MXene/Li scaffold to construct a composite anode with both ionic and electronic conductivity. Benefiting from the lithiophilic sites on the MXene surface and the curved pore geometry, this structure guides uniform lithium deposition within the internal cavities, while simultaneously buffering volume changes and suppressing dendrite growth. Moreover, the continuous charge-transport network accelerates reaction kinetics and enables compatibility with high-loading cathodes. Based on this strategy, the symmetric cell achieved stable cycling for over 1750 h; the LiFePO₄ (LFP) full cell retained 72.6% of its capacity after 1000 cycles at 1C; and the high-loading NCM90 pouch cell delivered an energy density of 392 Wh kg⁻¹. Furthermore, the projected energy density of a 13-layer stacked cell reached 561 Wh kg⁻¹, demonstrating a feasible route toward high-performance QSSLMBs.
Innovations
- Innovative structural design:
A “gel electrolyte–3D MXene/Li scaffold” integrated composite anode is constructed for the first time, enabling the synergistic coupling of dual ionic/electronic conduction networks. This design overcomes the limitations of the conventional separated electrode/electrolyte configuration and simultaneously achieves dynamic volume accommodation and efficient charge transport. - Innovative working mechanism:
The lithiophilic sites of the MXene scaffold reduce the lithium nucleation overpotential, while the hollow pore channels spatially confine lithium deposition and alleviate mechanical stress. The seamless integration of the PDOL gel electrolyte with the electrode forms a stable interface, and the high electronic conductivity of MXene, together with the ionic transport capability of the gel electrolyte, synergistically enhances reaction kinetics. - Practical breakthrough:
This strategy is compatible with high-loading high-nickel cathodes. The pouch cell exhibits record-high energy density and cycling stability among comparable reports, while also possessing flexibility and foldability. Therefore, it simultaneously fulfills the requirements of high energy density, long cycle life, and practical applicability.
Detailed Figure Interpretation
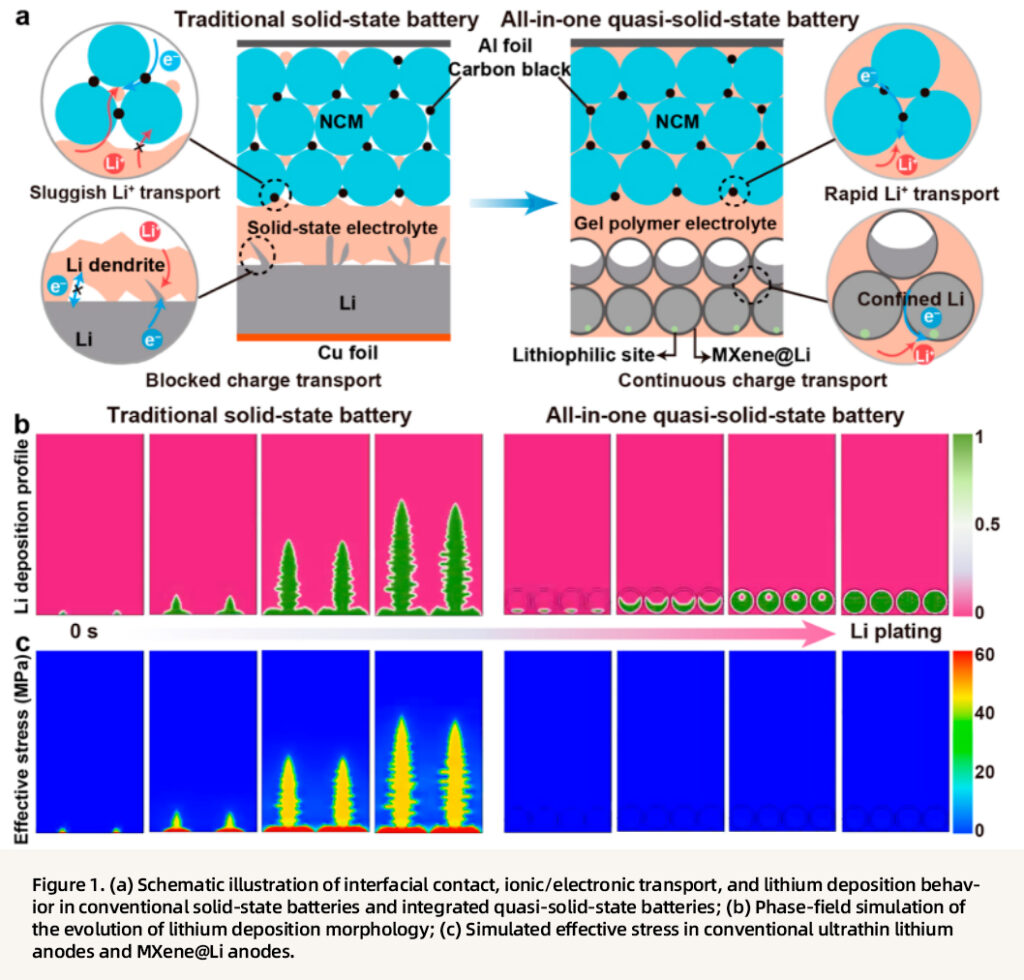
The core advantages of the integrated design are clearly illustrated. First, in conventional batteries, planar contact leads to sluggish ion transport, localized lithium deposition, severe dendrite growth, and concentrated interfacial stress, ultimately resulting in interfacial delamination and battery failure. In contrast, the integrated design employs the MXene@Li composite anode to provide continuous ion/electron transport pathways. The lithiophilic sites and pore-channel structure guide uniform lithium deposition into the internal cavities, thereby preventing dendrite formation. Meanwhile, the seamless fusion of the gel electrolyte with the electrode eliminates interfacial gaps, buffers volume change, and relieves mechanical stress. Furthermore, phase-field simulations confirm that lithium deposition in the integrated structure is much more uniform, and that the effective stress is significantly lower than that in the conventional configuration, validating the synergistic effects of stress regulation and dendrite suppression.
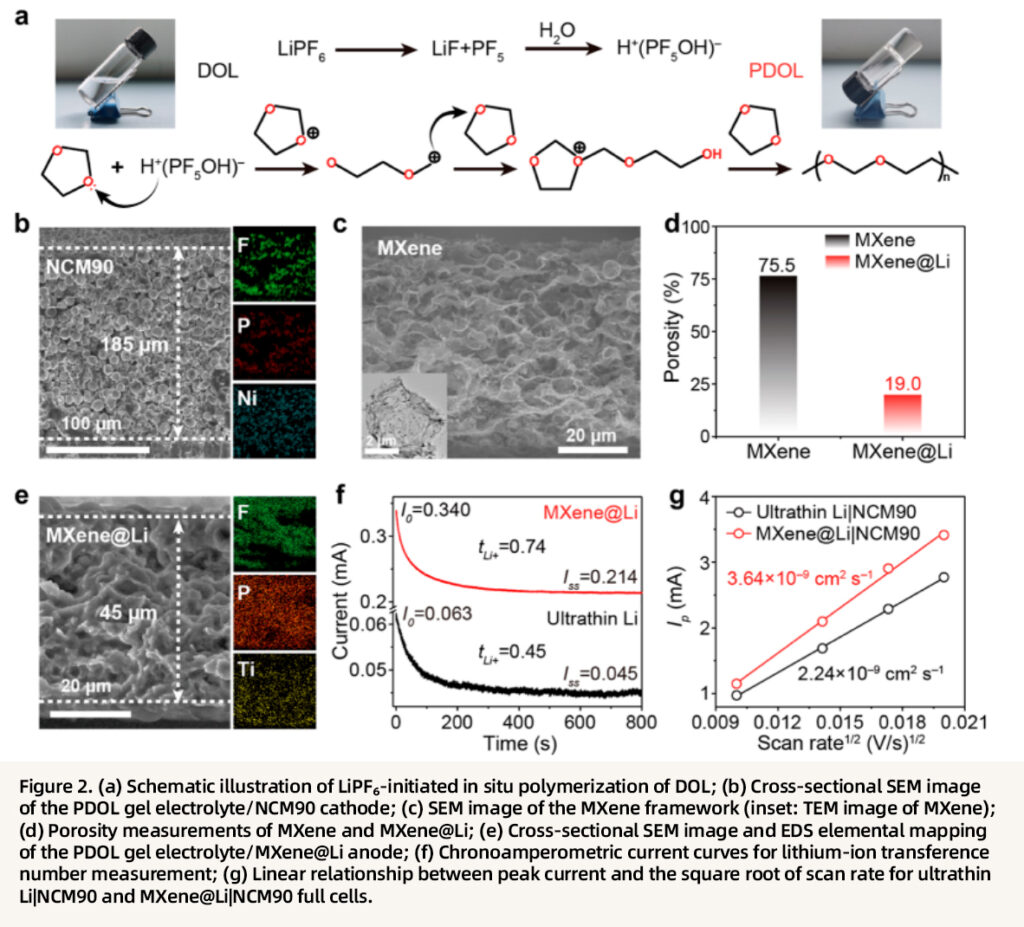
The construction and performance of the integrated structure are systematically verified. LiPF₆ decomposition generates acidic species that initiate the in situ ring-opening polymerization of DOL to form the PDOL gel electrolyte, which simultaneously infiltrates the MXene framework and NCM cathode, achieving seamless electrode–electrolyte integration. The MXene framework is modified with Ag nanoparticles to enhance lithiophilicity, and after lithium plating, an MXene@Li composite anode is obtained while retaining a porosity of 19.0%, thereby balancing ionic accessibility and structural stability. EDS mapping confirms the uniform distribution of F and P elements in both the MXene@Li anode and the NCM cathode, indicating sufficient infiltration of the gel electrolyte. The MXene@Li anode exhibits a lithium-ion transference number of 0.74, significantly higher than that of the ultrathin Li anode (0.45), and its lithium-ion diffusion coefficient (3.64 × 10⁻⁹ cm² s⁻¹) is twice that of the conventional structure. Linear sweep analysis further verifies that charge-transport kinetics are substantially improved in the integrated architecture, laying the foundation for efficient electrochemical reactions under high-loading conditions.
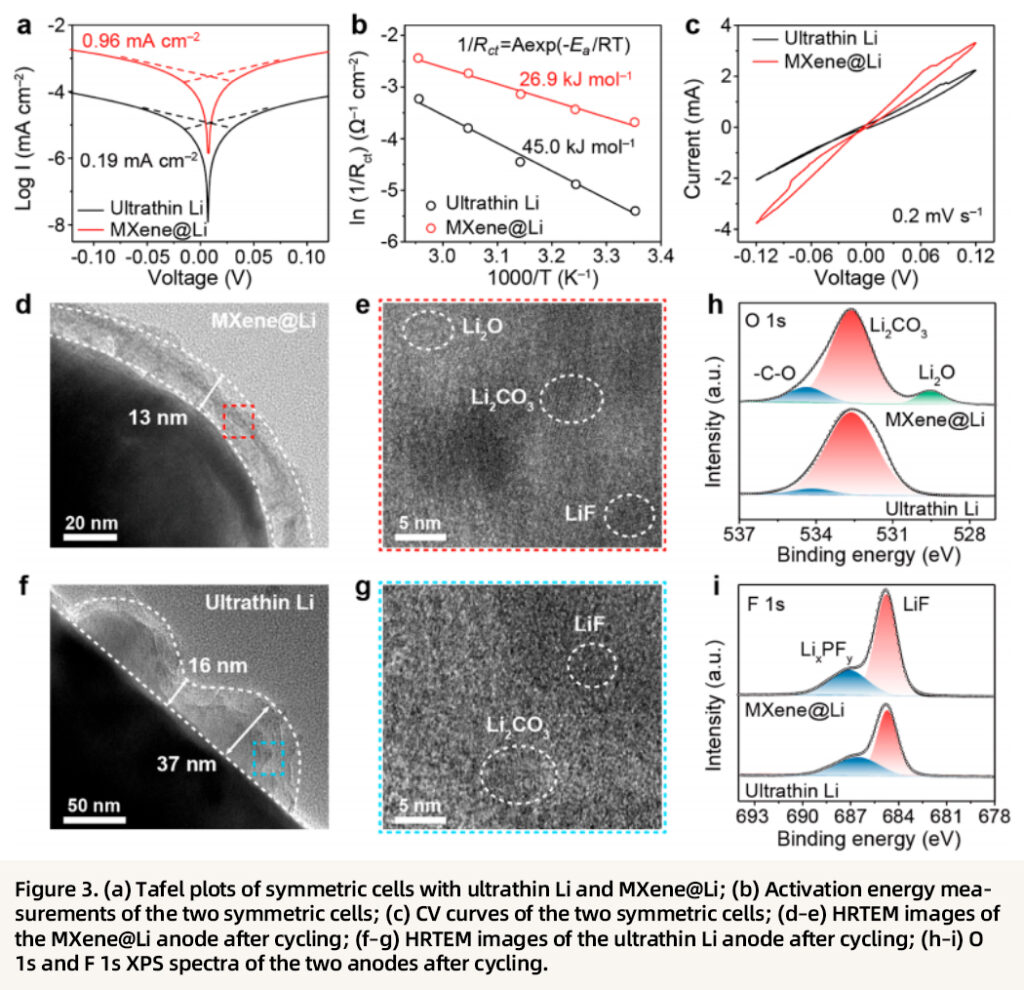
The kinetic advantages and interfacial stability are further elucidated in depth. The MXene@Li symmetric cell shows an exchange current density of 0.96 mA cm⁻², which is five times higher than that of the ultrathin Li cell (0.19 mA cm⁻²), while its activation energy (26.9 kJ mol⁻¹) is markedly lower than that of the ultrathin Li cell (45.0 kJ mol⁻¹), confirming lower interfacial charge-transfer resistance and more efficient ion transport. In addition, the MXene@Li anode forms a uniform mixed SEI layer rich in Li₂O, Li₂CO₃, and LiF, which is compact and ionically conductive. By contrast, the SEI layer on the ultrathin lithium metal anode is heterogeneous, contains a higher proportion of organic species, and exhibits greater ionic transport resistance. Correspondingly, the MXene@Li cell displays higher current response and smaller voltage hysteresis in CV measurements, indicating superior reversibility of lithium plating/stripping and enhanced interfacial stability.
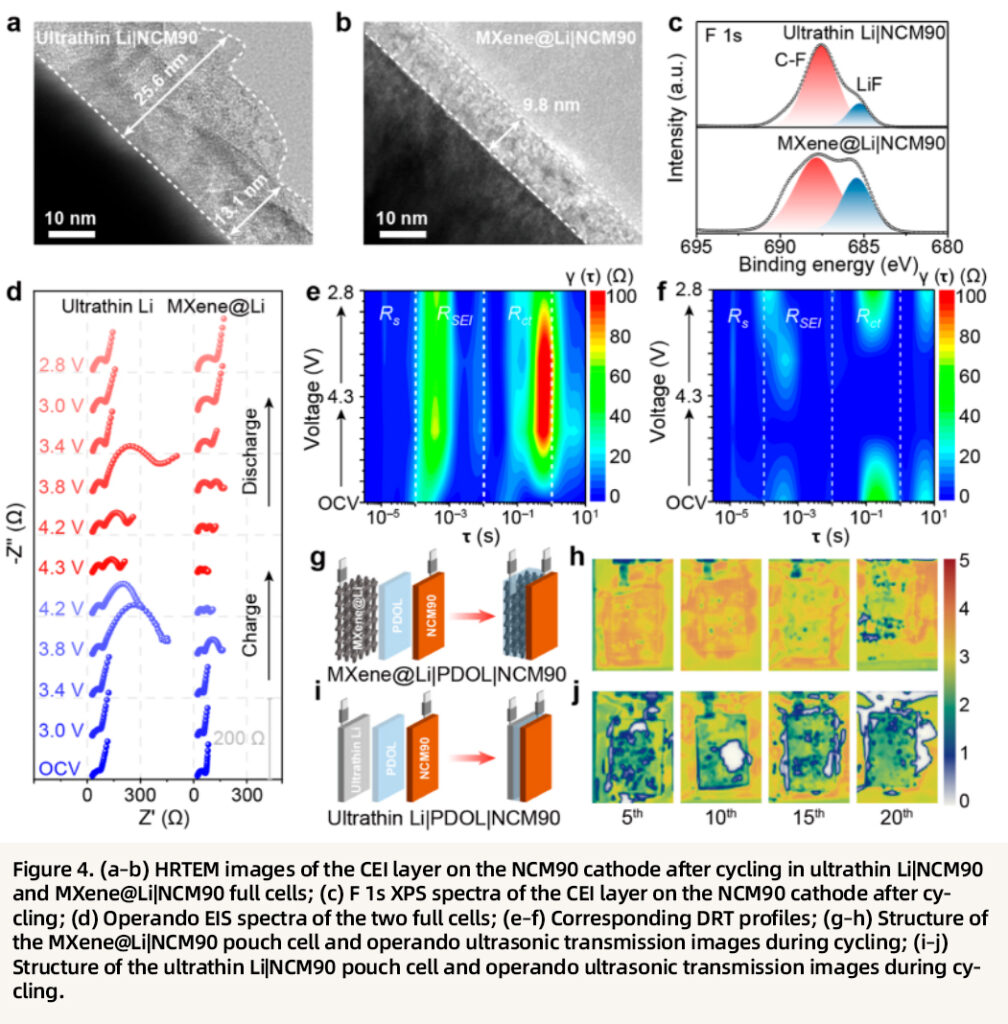
The interfacial stability of the integrated configuration is also directly visualized. In the MXene@Li system, the NCM cathode surface forms a thin and uniform CEI layer (~10 nm) enriched with inorganic species such as LiF and Li₃PO₄, thereby providing effective protection. In contrast, the CEI layer in the ultrathin Li system is thicker and more heterogeneous (~30 nm), with a higher fraction of organic components and inferior structural stability. Operando EIS and DRT analyses further reveal that both the SEI resistance and charge-transfer resistance of the MXene@Li full cell remain consistently lower than those of the ultrathin Li counterpart, demonstrating that the integrated structure can maintain long-term stable interfacial contact and efficient charge transport. Moreover, operando ultrasonic imaging shows that the MXene@Li pouch cell preserves intimate interfacial contact throughout cycling without obvious gas generation or void formation, whereas the ultrathin Li pouch cell suffers from severe interfacial detachment and intense side reactions.
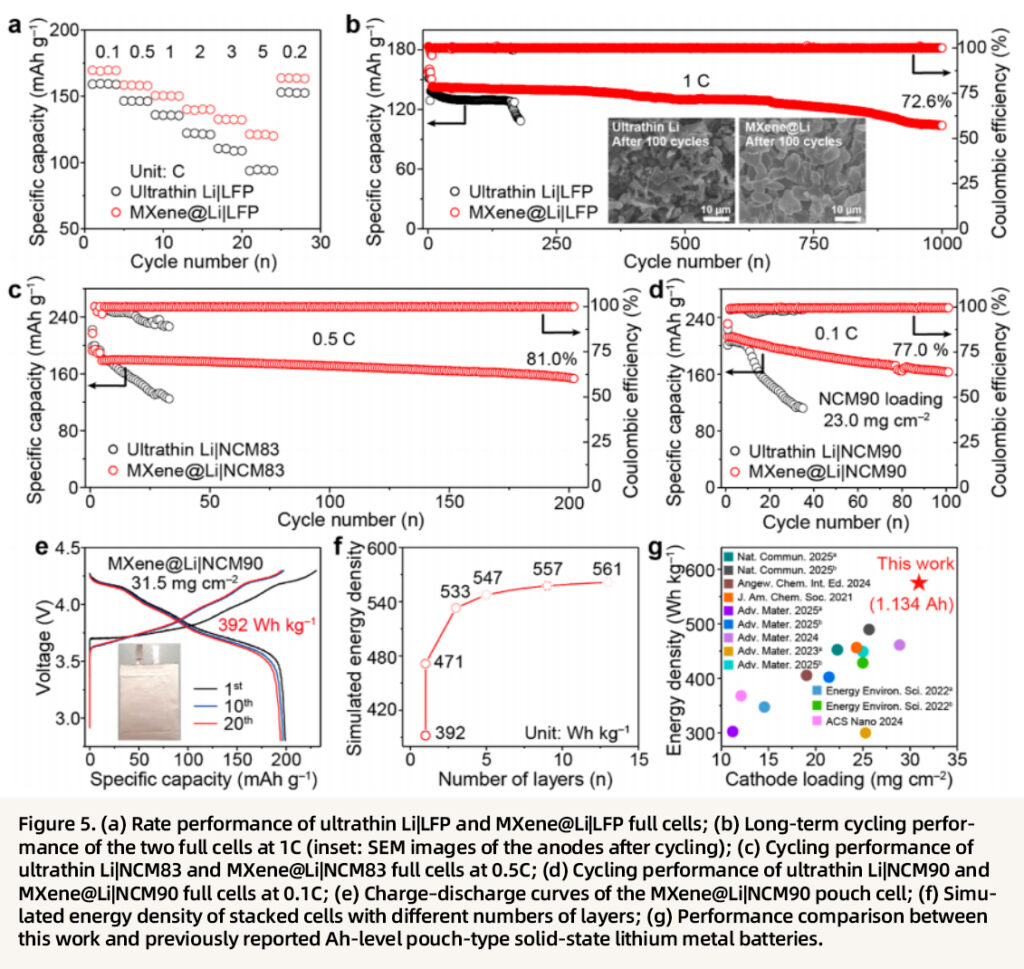
The practical electrochemical performance of the batteries is comprehensively demonstrated. The MXene@Li|LFP full cell delivers excellent rate capability over a wide range of 0.1–5 C and retains 72.6% of its capacity after 1000 cycles at 1C. Even after long-term cycling, the anode remains smooth and dense without dendrite formation, whereas severe dendrites and significant capacity decay are observed in the ultrathin Li cell. When paired with high-loading NCM83/NCM90 cathodes, MXene@Li exhibits far superior cycling stability and capacity output compared with the conventional system. In particular, the full cell with an NCM90 cathode loading of 23.0 mg cm⁻² achieves an initial areal capacity of 5.39 mAh cm⁻² and still retains 3.78 mAh cm⁻² after 100 cycles. For pouch-cell demonstration, the NCM90 pouch cell with a cathode loading of 31.5 mg cm⁻² achieves an energy density of 392 Wh kg⁻¹ and also exhibits flexible and foldable characteristics. The projected energy density of a 13-layer stacked cell reaches 561 Wh kg⁻¹, outperforming most previously reported pouch-type solid-state lithium metal batteries.
Conclusion
In summary, this work successfully addresses the interfacial stability and charge-transport bottlenecks in quasi-solid-state lithium metal batteries through an ion/electron mixed-conducting gel electrolyte–anode integrated strategy. The key mechanism lies in the integrated composite anode formed by a three-dimensional hollow MXene/Li scaffold and PDOL gel electrolyte, which simultaneously provides dual ionic/electronic conductivity. Specifically, the lithiophilic sites of MXene reduce the lithium nucleation overpotential, while the hollow pore channels spatially confine lithium deposition, buffer volume changes, and suppress dendrite growth and stress accumulation. Meanwhile, the seamless fusion of the PDOL gel with the electrode establishes a stable interface and provides efficient ion transport pathways. The continuous charge-transport network, together with compatibility with high-loading cathodes, further accelerates reaction kinetics. Electrochemical tests demonstrate that this integrated structure enables stable symmetric-cell cycling for over 1750 h, achieves a 72.6% capacity retention for the LiFePO₄ (LFP) full cell after 1000 cycles at 1C, and delivers an energy density of 392 Wh kg⁻¹ for a high-loading NCM90 pouch cell. This design effectively breaks the conventional trade-off between energy density and cycling life, simultaneously offering high energy density, long-term cycling stability, and mechanical flexibility. Therefore, it provides a new paradigm for the practical development of next-generation high-performance quasi-solid-state lithium metal batteries and advances lithium metal batteries toward high-energy-density energy-storage applications.
