Background
Solid polymer electrolytes (SPEs) have shown great potential in lithium metal batteries due to their high safety, flexibility, and good processability. However, unstable interfaces between the lithium metal anode and SPEs, lithium dendrite growth, and side reactions have severely hindered their commercial application. For research related to lithium metal anodes, Xnergy’s Metallic Lithium Series and Lithium Alloy & Pre-Lithiation Solutions may provide useful material options.
Article Overview
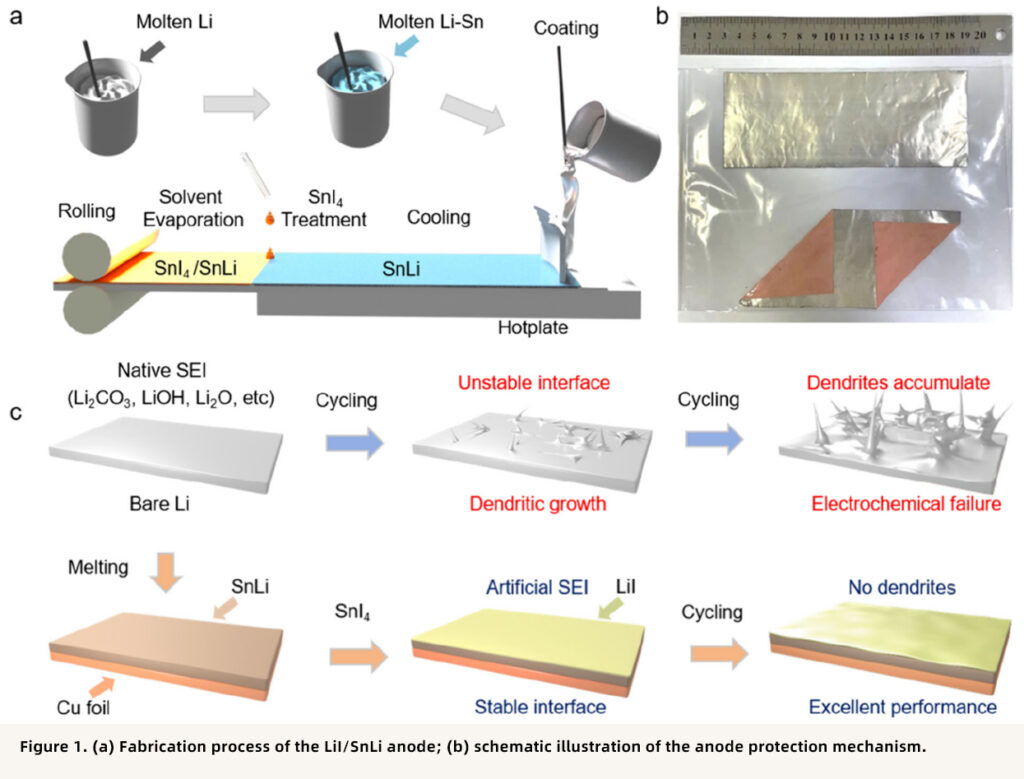
This study proposes a novel LiI-coated SnLi alloy composite anode (LiI/SnLi). A Li-Sn alloy was first prepared via a melting method, followed by a surface reaction to generate a LiI-rich artificial SEI layer. This structure not only provides abundant lithium deposition sites to suppress dendrite growth, but also effectively reduces side reactions between the SPE and lithium metal, significantly improving the cycling stability of the battery. For related lithium alloy research, Xnergy’s Lithium Alloy & Pre-Lithiation Solutions can be referenced.
Key Points
Point 1: Synergistic design of the SnLi alloy and LiI artificial SEI
By alloying Sn with Li through a melting method, a Li₂₂Sn₅ alloy anode with high binding energy was prepared, which significantly improved the wettability of molten Li on Cu foil. Further treatment with SnI₄ generated an in situ LiI-rich layer on the alloy surface, forming an artificial SEI that is electronically insulating but ionically conductive, thereby effectively suppressing interfacial side reactions and lithium dendrite growth. If your work involves lithium metal or lithium-alloy anode design, Xnergy’s Metallic Lithium Series and Lithium Alloy & Pre-Lithiation Solutions may be relevant.
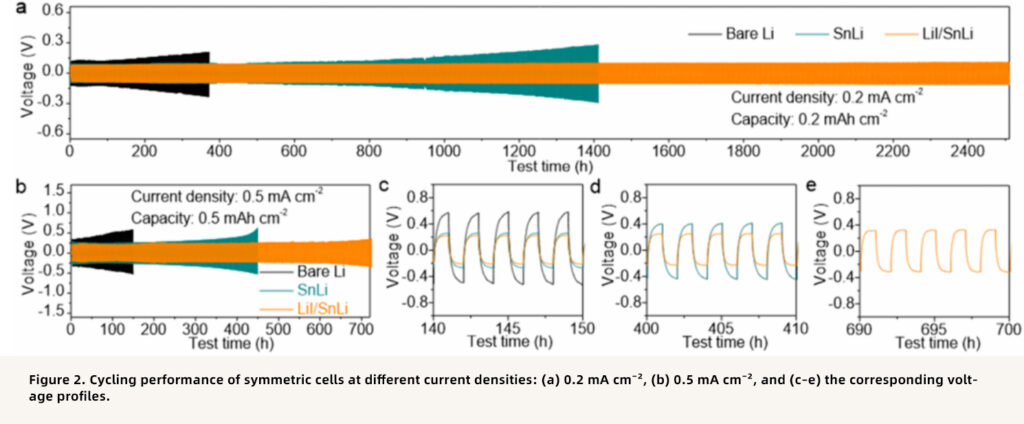
Point 2: Greatly improved interfacial stability, with symmetric cells cycling for over 2500 hours
The LiI/SnLi anode exhibited excellent interfacial stability in a PVDF-based SPE. Symmetric cells operated stably for over 2500 hours at 0.2 mA cm⁻² with a low overpotential of only 110 mV, far superior to bare Li, which short-circuited after about 370 hours, and SnLi, which failed after 1400 hours. Even at a higher current density of 0.5 mA cm⁻², the cell still maintained stable operation for 700 hours.
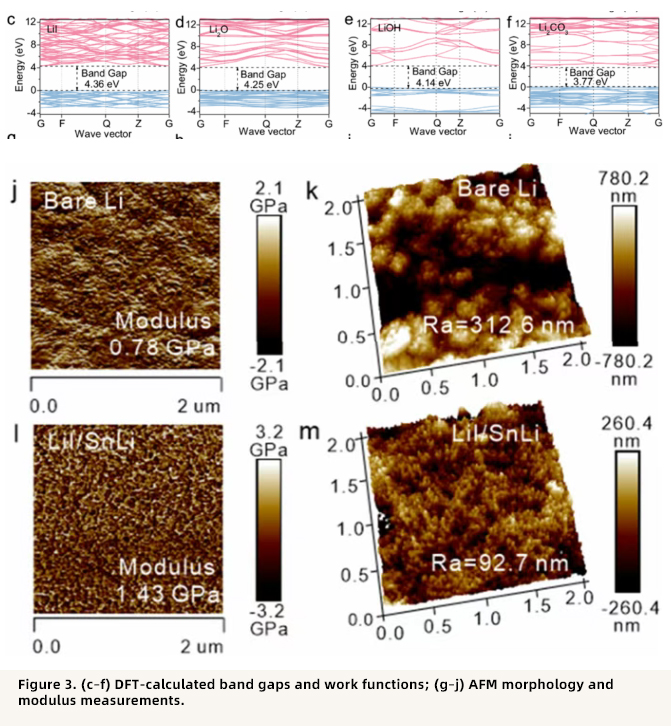
Point 3: Electronic insulation and mechanical stability of the artificial SEI layer
DFT calculations showed that LiI has a relatively high band gap (4.36 eV) and work function (5.72 eV), outperforming common SEI components such as Li₂O, LiOH, and Li₂CO₃, thus effectively blocking electron penetration and suppressing electrolyte decomposition. AFM measurements further revealed that the LiI layer has a higher Young’s modulus (1.43 GPa) and lower surface roughness (92.7 nm), which helps promote uniform lithium deposition.
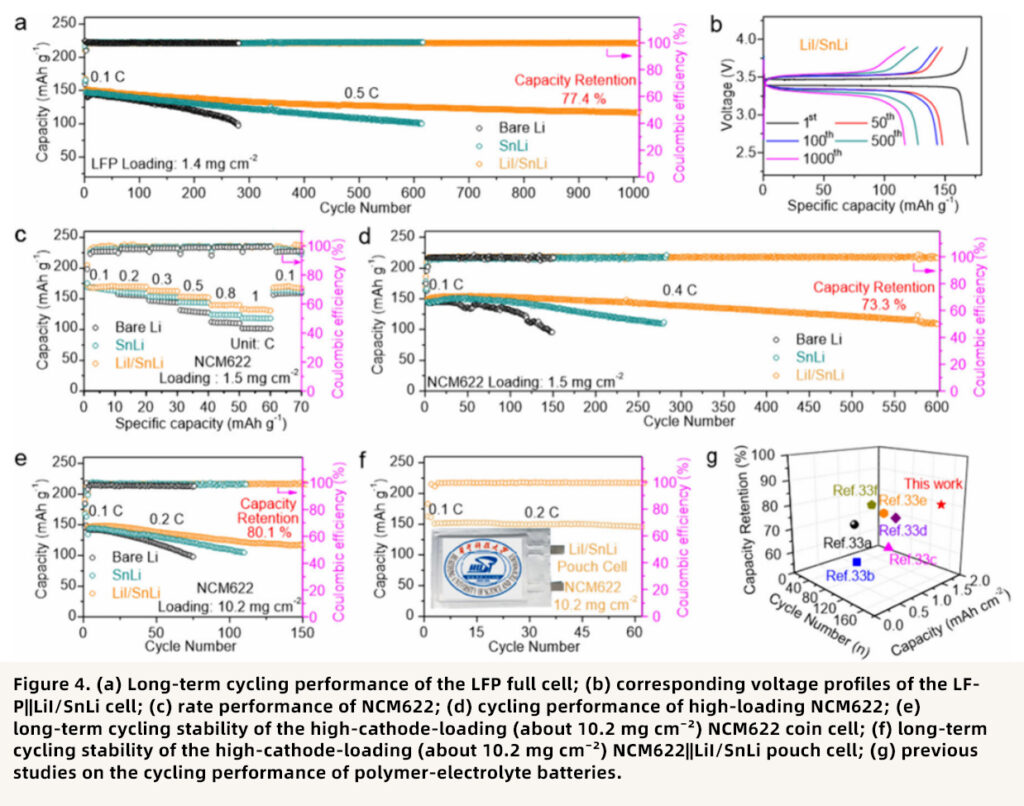
Point 4: Excellent full-cell performance, compatible with high-loading cathodes and pouch cells
The LFP full cell based on the LiI/SnLi anode retained 77.4% of its capacity after 1000 cycles at 0.5C. For researchers interested in LFP-based cell assembly, Xnergy’s LFP Cathode Sheet may be a useful option. The NCM622 full cell maintained 73.3% of its capacity after 600 cycles. Even under a high NCM622 loading of 10.2 mg cm⁻² and an N/P ratio of 4.8, the cell still cycled stably for 150 cycles. For high-loading NCM622 and pouch-cell-related studies, Xnergy’s NCM622 / Lithium Metal Pouch Battery Dry Cell and the broader Dry Pouch Cell category may also be relevant. In addition, the pouch cell showed good interfacial uniformity in ultrasonic imaging and remained safe during cutting tests.