LiNbOCl4 (LNOC) Solid Electrolyte Powder
LiNbOCl₄ · Niobium-Based Oxychloride · Soft Oxyhalide Superionic Conductor
A niobium-based oxychloride solid electrolyte for high-voltage all-solid-state battery research, with high room-temperature Li⁺ conductivity and a low migration barrier — a leading member of the soft oxyhalide superionic conductor family. Available from Xnergy Materials as a U.S.-based supplier with global shipping for academic and industrial research customers.

LiNbOCl4 (chemical formula LiNbOCl4, abbreviated LNOC) is a niobium oxychloride solid electrolyte for all-solid-state battery research. Xnergy's research-grade LiNbOCl4 powder exhibits a room-temperature ionic conductivity of approximately 7 mS/cm, a Li⁺ migration activation energy of 0.256 eV, and demonstrated stability with high-voltage cathodes including LCO at 4.6 V (96.7% Coulombic efficiency) and Ni83. In stock for purchase at Xnergy Materials — a direct U.S. supplier of LiNbOCl4 powder — with a 10 g minimum order quantity. Bulk pricing, custom particle sizes, custom batch quantities, and joint research collaborations are supported on request.
A Niobium-Based Soft Oxyhalide Superionic Conductor
LiNbOCl4, abbreviated LNOC, belongs to the recently discovered family of soft oxyhalide solid electrolytes, first reported in the LiMOCl4 (M = Nb, Ta) system by Tanaka and co-workers. These materials combine the high oxidation stability of halide-based solid electrolytes with the fast Li⁺ transport associated with mixed oxygen–chlorine anion frameworks. Within this family, LiNbOCl4 has emerged as one of the most highly conducting members yet characterized.
The high ionic conductivity of LNOC arises from a Li-disordered tetragonal crystal structure consisting of isolated 1-D [NbOCl4]− anionic chains with energetically favorable orientational disorder. This disorder couples to multiple equi-energetic Li⁺ sites in the lattice, producing a soft, flexible framework that supports fast Li⁺ migration even in cold-pressed powder form — a property critical for practical all-solid-state battery (ASSB) cell assembly.
Xnergy Materials supplies research-grade LiNbOCl4 powder for academic and industrial laboratories developing next-generation cold-pressed solid-state cells, particularly those targeting high-voltage layered oxide cathode pairing. The material is vacuum-sealed under argon, in stock, and ships within one week of order confirmation.
The Case for LiNbOCl4 in ASSB Research
Niobium oxychloride solid electrolytes — and LiNbOCl4 in particular — occupy a distinctive position in the broader solid electrolyte landscape. As a niobium oxychloride material, LNOC combines the high room-temperature ionic conductivity typical of leading sulfide systems with the wide electrochemical window of halide chemistries, while remaining cold-press compatible without high-temperature sintering. Among the soft oxyhalide superionic conductors reported to date, LiNbOCl4 exemplifies this trade-off resolution and has emerged as one of the most actively studied niobium oxychloride compositions in the all-solid-state battery research community.
Niobium chemistry: familiar, abundant, scalable
Niobium has a well-established role in lithium battery research as a coating constituent (LiNbO3 is a standard cathode-coating chemistry for high-nickel NCM). Compared with tantalum, niobium offers significantly greater natural abundance and lower raw-material cost, which makes Nb-based oxyhalides such as LNOC attractive for research programs concerned with eventual scale-up. From a chemical standpoint, Nb(V) and Ta(V) form analogous oxychlor complexes, so LiNbOCl4 and LiTaOCl4 are widely used as parallel research samples for studying central-metal effects in oxyhalide electrolytes.
High-voltage cathode compatibility
One of the principal limitations of sulfide solid electrolytes such as LPSC and LGPS is their narrow oxidation stability against high-voltage layered oxide cathodes. LiNbOCl4, with its mixed-anion framework and oxychloride character, demonstrates stable interfaces with both LiCoO2 at 4.6 V and high-nickel NCM (Ni83) cathodes — making it a useful candidate for high-energy-density ASSB studies.
Cold-press compatibility for cell fabrication
LNOC retains its high ionic conductivity in cold-pressed powder form, enabling solid-state cell assembly at room temperature without densification at elevated temperature. This is a key practical advantage relative to garnet-type or NASICON-type oxide solid electrolytes, which generally require high-temperature sintering to reach acceptable grain-boundary contact.
Three Solid Electrolytes for Comparative Research
Xnergy Materials currently supplies three members of the lithium oxychloride solid electrolyte family, enabling direct LNOC vs LTOC comparison studies as well as broader research into how central-metal substitution (Nb vs Ta) and lithium stoichiometry (Li:M ratio) influence ionic transport, microstructure, and cathode interfacial behavior in oxyhalide systems. For research groups designing parallel LNOC vs LTOC experiments under identical cell architectures, all three products are available in stock from a single supplier.
LiNbOCl4
Niobium-based oxyhalide superionic conductor
Specifications below ↓LiTaOCl4
Tantalum-based oxychloride, parallel chemistry to LNOC
View product →Li2TaOCl6
Lithium-rich Li:Ta = 2:1 stoichiometry
View product →Researchers studying polyanion structure, Li⁺ migration pathways, and composition–property relationships in lithium-ion superionic conductors frequently acquire two or all three of these products together. Common comparison axes include: Nb vs Ta central-metal effects (LNOC vs LiTaOCl4, often searched as "LNOC vs LTOC"), lithium stoichiometry effects (LiTaOCl4 vs Li2TaOCl6), and broad oxyhalide chemistry benchmarking against sulfide and oxide systems.
LNOC Across Four Chemistry Families
Solid electrolytes are typically grouped into four chemistry families. Each comes with its own performance trade-offs. LiNbOCl4 sits in the soft oxyhalide branch of halide-derived electrolytes and combines characteristics of both halide and oxide chemistries.
| Family | Representatives | Conductivity (RT) | Moisture Tolerance | High-V Cathode Stability | Cold-Press Friendly |
|---|---|---|---|---|---|
| Sulfide | LPSC, LGPS, Li6PS5Cl | 10+ mS/cm | Poor (releases H₂S) | Limited | Good |
| Oxide (garnet, NASICON) | LLZO, LATP | 0.1–1 mS/cm | Excellent | Good | Requires sintering |
| Halide (chloride) | Li₃YCl₆, Li₃InCl₆, LZC | 1–3 mS/cm | Moderate | Good | Good |
| Oxyhalide / Oxychloride | LiNbOCl4, LiTaOCl4, Li2TaOCl6 | 6–11 mS/cm | Improved vs sulfide | Excellent (4.6 V validated) | Yes (cold-press) |
The oxyhalide row reflects literature-reported ranges for the LiMOCl4 family. Reported room-temperature conductivities for optimized LiNbOCl4 have reached ~10–11 mS/cm under specific synthesis and processing conditions (Newnham et al., JACS 2024; Tanaka et al., 2023), placing the family at the high end of all currently characterized lithium-ion solid electrolytes.
Representative Electrochemical Data
The figure below shows representative measurements taken on Xnergy's research-grade LiNbOCl4 powder, including impedance spectroscopy (Nyquist plot), X-ray diffraction phase identification, variable-temperature Arrhenius analysis (Eₐ = 0.256 eV), and charge–discharge curves of full all-solid-state cells with LCO at 4.6 V and Ni83 high-nickel layered oxide cathodes. The data reflect typical batch performance; per-batch characterization reports (XRD, EIS, particle-size analysis) are available with technical data sheets on request.
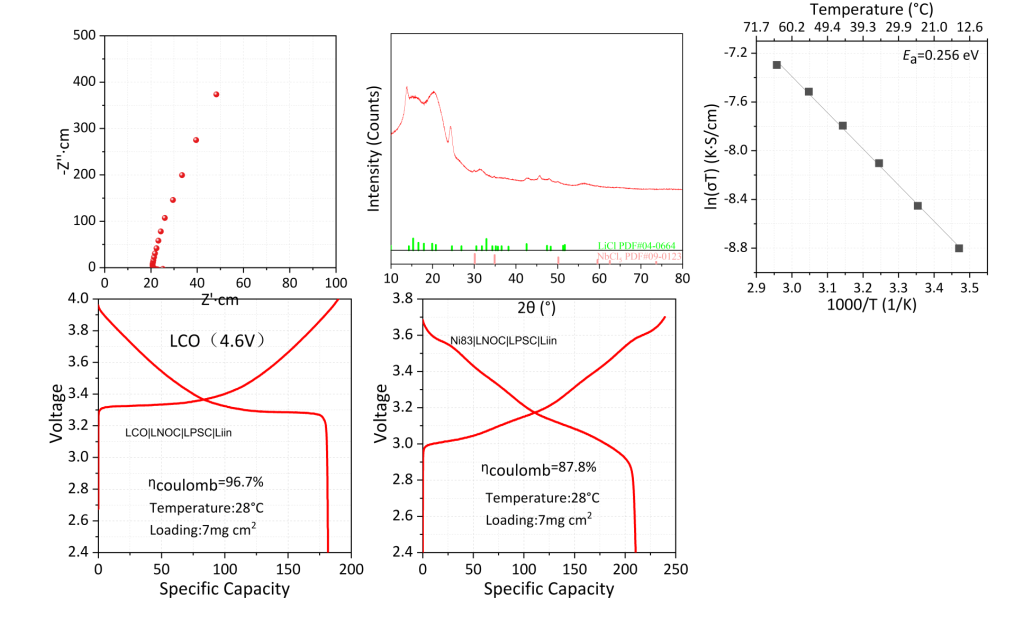
Electrochemical & Structural Data Summary
Top row, left → right:
Nyquist plot from room-temperature impedance spectroscopy showing low interfacial resistance;
X-ray diffraction pattern confirming the LiNbOCl4 crystalline phase;
Arrhenius plot of ln(σT) vs 1000/T yielding a Li⁺ migration activation energy of 0.256 eV.
Bottom row, left → right:
Charge–discharge curve of an LCO | LNOC | LPSC | Li-In cold-pressed all-solid-state cell at 28 °C, 7 mg/cm² loading, with 96.7% first-cycle Coulombic efficiency at a 4.6 V upper cutoff;
Charge–discharge curve of a Ni83 | LNOC | LPSC | Li-In cell with 87.8% first-cycle Coulombic efficiency, demonstrating compatibility with high-nickel layered cathode chemistries.
Material Properties & Packaging
Where LiNbOCl4 Fits in Battery R&D
LiNbOCl4 is intended primarily for advanced solid-state battery research at universities, national laboratories, and corporate R&D programs. Typical research applications for LNOC powder include:
Researchers comparing niobium oxychloride electrolytes against established sulfide systems (LPSC, LGPS) or oxide systems (LLZO, LATP) often pair LiNbOCl4 with our other in-stock solid-state electrolytes for direct head-to-head benchmarking under identical cell architectures. Typical experimental endpoints include interfacial impedance measurements, composite cathode fabrication using LNOC as a catholyte, electrochemical stability window determination, and Li⁺ migration pathway analysis. The Xnergy oxychloride family — LNOC, LiTaOCl4, and Li2TaOCl6 — additionally enables controlled studies of how central-metal substitution and lithium stoichiometry alter Li⁺ transport within the same broad chemistry, supporting the next generation of lithium-ion superionic conductor research.
Beyond Standard Pack Sizes
For projects whose requirements exceed our standard pack sizes, or that require composition or particle-size adjustments, Xnergy Materials offers extended supply, bulk purchase agreements, and joint development services for LiNbOCl4 (LNOC) and the wider niobium oxychloride product family. We work directly with research teams as a direct supplier to align material specifications with experimental needs, and we typically return quotes for bulk orders within one to two business days.
Custom Synthesis, Bulk Purchase & Long-Term Supply
Long-term supply agreements, bulk LiNbOCl4 purchase contracts, and collaborative R&D partnerships are welcomed. Whether you are running a multi-year academic program or a corporate ASSB development effort, our team can support your sourcing roadmap for LiNbOCl4 (LNOC) and other niobium oxychloride solid electrolytes.
- Custom particle size distributions (D₅₀ tailoring)
- Custom batch sizes from 100 g to multi-kilogram pilot lots
- Stoichiometry adjustments within the Li-Nb-O-Cl system
- Joint development agreements & long-term supply contracts
- Material Transfer Agreements (MTAs) for academic programs
- Per-batch characterization reports (XRD, EIS, particle-size analysis)
sales@xnergy.us · 1-530-433-0971
Frequently Asked Questions
What is LiNbOCl4 (LNOC)?
LiNbOCl4 — abbreviated LNOC — is a niobium-based oxychloride solid electrolyte and one of the most highly conducting members of the recently discovered soft oxyhalide superionic conductor family. It was first reported in the LiMOCl4 (M = Nb, Ta) system by Tanaka et al., with subsequent JACS work characterizing its disordered tetragonal framework and Li⁺ migration pathways. LNOC is supplied by Xnergy as a research-grade powder for all-solid-state battery (ASSB) studies.
Where can I buy LiNbOCl4 powder?
Xnergy Materials supplies LiNbOCl4 (LNOC) powder for research and R&D applications. Standard pack sizes start at a 10 g minimum order quantity, with 25 g and 50 g packs available, and larger custom batches by quote. The material is in stock and typically ships within one week of order confirmation. Request a quote →
What is the ionic conductivity of LiNbOCl4?
Xnergy's research-grade LiNbOCl4 exhibits a room-temperature ionic conductivity of approximately 6–8 mS/cm, with a low Li⁺ migration activation energy of 0.256 eV. Literature reports for optimized LiNbOCl4 have reached ~10–11 mS/cm under specific synthesis conditions, placing the material among the highest-conducting oxyhalide solid electrolytes characterized to date.
What is the difference between LNOC (LiNbOCl4) and LTOC (LiTaOCl4)?
LNOC and LTOC are sister materials within the LiMOCl4 (M = Nb, Ta) oxyhalide family, sharing similar polyanion chemistry, comparable ionic conductivities, and similar high-voltage cathode compatibility. The principal differences are the central metal element — niobium for LNOC vs tantalum for LTOC — which influences crystal structure details, raw-material economics (Nb is more abundant), and certain interfacial behavior. Xnergy supplies both products to enable direct comparative research.
Is LiNbOCl4 stable against high-voltage cathodes such as LCO at 4.6 V and Ni83?
Yes. LiNbOCl4 has been validated in cold-pressed all-solid-state cells with LCO cathodes operating up to 4.6 V (96.7% first-cycle Coulombic efficiency at 28 °C, 7 mg/cm² loading) and with Ni83 high-nickel layered oxide cathodes (87.8% first-cycle Coulombic efficiency). LNOC's wide electrochemical window makes it suitable for high-voltage cathode pairing studies that exceed the stability range of typical sulfide solid electrolytes.
Is LiNbOCl4 moisture sensitive?
LiNbOCl4 has improved moisture tolerance compared with sulfide solid electrolytes such as LPSC and LGPS, but it should still be stored and handled under inert atmosphere (argon-filled glovebox preferred). Brief air exposure should be avoided to preserve ionic conductivity. Xnergy ships LNOC vacuum-sealed under argon for research handling.
Does LiNbOCl4 work with cold-press assembly?
Yes. Soft oxyhalide solid electrolytes including LiNbOCl4 are designed for cold-pressed cell fabrication and exhibit high relative density at room temperature without high-temperature sintering. This is one of LNOC's key advantages over oxide-based solid electrolytes such as LLZO that require densification at elevated temperatures.
What is the activation energy for Li⁺ conduction in LiNbOCl4?
The Li⁺ migration activation energy in LiNbOCl4 measured on Xnergy's research-grade powder is 0.256 eV, determined from variable-temperature impedance spectroscopy across approximately 12 °C to 72 °C. This low barrier is consistent with the high room-temperature ionic conductivity and supports the picture of orientationally disordered [NbOCl4]− chains enabling fast Li⁺ transport.
What is the minimum order quantity for LNOC?
The minimum order quantity is 10 grams. Standard pack sizes are 10 g, 25 g, and 50 g. For larger research batches, multi-hundred-gram, or kilogram-scale pilot quantities, please contact us with your specifications and we will return a per-batch quote.
Does Xnergy offer custom particle sizes, custom synthesis, or joint development for LiNbOCl4?
Yes. Xnergy supports custom particle size distributions, custom batch sizes, and stoichiometry adjustments within the Li-Nb-O-Cl system for research programs studying composition–property relationships. We also welcome long-term supply agreements, joint development collaborations, and Material Transfer Agreements (MTAs) with academic and industrial partners working on next-generation oxyhalide and oxychloride solid electrolytes. Contact sales@xnergy.us to discuss your project.
How does LNOC compare with sulfide solid electrolytes such as LPSC?
LiNbOCl4 matches the ionic conductivity range of leading sulfide solid electrolytes such as Li6PS5Cl (LPSC), while offering meaningfully better moisture tolerance and a wider electrochemical stability window suitable for high-voltage layered oxide cathodes operating above 4.5 V. Sulfides retain advantages in raw material cost and very high room-temperature conductivity, but LNOC is often preferred for high-voltage ASSB cathode-pairing studies.
Is LiNbOCl4 available for purchase internationally?
Yes. Xnergy Materials is a U.S.-based supplier of LiNbOCl4 (LNOC) powder and ships internationally to academic and industrial research customers. International orders are supported with appropriate export and customs documentation. Standard pack sizes (10 g, 25 g, 50 g) and bulk research batches are both available for purchase. Contact sales@xnergy.us for an international shipping quote.
What are the key differences between LNOC and LTOC for solid-state battery research?
The principal LNOC vs LTOC differences for solid-state battery research are:
(1) Central metal element — niobium (Nb) for LNOC, tantalum (Ta) for LTOC;
(2) Raw-material economics — Nb is roughly an order of magnitude more abundant than Ta and meaningfully cheaper;
(3) Crystal-structure details — both materials adopt the LiMOCl4 framework but exhibit subtly different bond lengths and disorder profiles;
(4) Interfacial behavior — research has shown small but measurable differences in cathode interface formation.
Bulk performance metrics — room-temperature ionic conductivity (6–8 mS/cm), activation energy, and high-voltage cathode compatibility — are very similar. Many research groups acquire both LNOC and LTOC together to run controlled LNOC vs LTOC comparison studies under identical cell architectures. View LiTaOCl4 (LTOC) →
Other Solid Electrolytes from Xnergy
Researchers often pair LiNbOCl4 with other solid electrolyte families for benchmarking studies. The following materials are also stocked at Xnergy:
Selected Literature on LiNbOCl4 & Soft Oxyhalides
For researchers new to the oxyhalide family, the following references provide background on LiNbOCl4 structure, ion-transport mechanisms, and synthesis strategies:
- Newnham, J. A. et al. (2024). Critical Role of Framework Flexibility and Disorder in Driving High Ionic Conductivity in LiNbOCl4. Journal of the American Chemical Society.
- Tanaka, Y. et al. (2023). New Oxyhalide Solid Electrolytes with High Lithium Ionic Conductivity >10 mS/cm for All-Solid-State Batteries. (LiTaOCl4: 12.4 mS/cm; LiNbOCl4: 10.7 mS/cm.)
- Yu, T. et al. (2023). A family of oxychloride amorphous solid electrolytes for long-cycling all-solid-state lithium batteries. Nature Communications.
- Jeon, S. et al. (2025). Hydrochloric acid-free synthesis of LiNbOCl4 superionic conductor for all-solid-state Li batteries. Solid State Ionics.
- Newnham, J. A. et al. (2024). Origin of Fast Li⁺-Ion Conductivity in the Compressible Oxyhalide LiNbOCl4. Energy Storage Materials.
- Recent reviews on chloride and oxyhalide solid electrolytes — ACS Energy Letters, 2025.
- Solid-state battery — Wikipedia overview.
References listed for general scientific background. Per-batch characterization reports specific to Xnergy LiNbOCl4 material lots are available with technical data sheets upon request.
