
LZC (Li₂ZrCl₆)-Halide Solid-State Electrolyte
LZC Halide Solid-State Electrolyte (Li₂ZrCl₆) — chloride-based halide electrolyte with ~0.3 mS/cm ionic conductivity at 28 °C. Stable against 4 V-class cathodes (LCO, Ni83) without coatings. Validated in full all-solid-state cells delivering ~150 mAh/g (LCO) and ~190 mAh/g (Ni83). Cost-effective alternative to Li₃YCl₆.
Need a bulk order? Request a competitive quote today!
Name: LZC Halide Solid-State Electrolyte (Li₂ZrCl₆)
Material Form: Crystalline powder
Product Code: XN-LZC
Description:
LZC (Li₂ZrCl₆) is a chloride-based halide solid-state electrolyte for next-generation all-solid-state lithium batteries (ASSLBs). As a zirconium-based halide system, LZC offers a low-cost alternative to yttrium-based halide electrolytes (Li₃YCl₆) while delivering 0.3 mS/cm room-temperature ionic conductivity and intrinsic stability against high-voltage cathodes — eliminating the need for cathode interface coatings required by sulfide electrolyte systems.
Application:
Catholyte for all-solid-state lithium batteries. Suitable for sulfide-halide composite electrolyte architectures (paired with LPSC or LGPS as anode-side separator), high-voltage cathodes (LCO, NCM/Ni83), solid-state pouch cell prototyping, and halide electrolyte fundamental studies.
Specifications:
| Property | Value |
|---|---|
| Composition | Li₂ZrCl₆ |
| Ionic Conductivity | ~0.3 mS/cm @ 28 °C |
| Test Condition | EIS, 1.23 mm pellet, 28 °C |
| Crystal Structure | Li₂ZrCl₆ phase confirmed by XRD |
| Electrochemical Window | Stable vs 4 V-class cathodes |
| Form | Crystalline powder |
| Storage | Inert atmosphere (Ar / N₂), sealed, moisture-excluded |
Values measured by Xnergy. Typical values for reference; not guaranteed unless otherwise specified.
Characteristics:
High-voltage cathode compatibility
Halide chemistry is intrinsically stable against 4 V-class cathodes (LCO, Ni83). No LiNbO₃ or other interface coatings required, simplifying cell architecture and reducing cathode interface resistance.
Cost-effective halide chemistry
Zirconium-based composition offers a lower-cost alternative to yttrium-based halide electrolytes (Li₃YCl₆), supporting commercially viable solid-state battery scale-up.
Validated in full ASSLB cells
LCO|LZC|LPSC|Li-In: ~150 mAh/g, 97.24% coulombic efficiency. Ni83|LZC|LPSC|Li-In: ~190 mAh/g, 88.64% coulombic efficiency. Both at 7 mg/cm² loading, 28 °C.
Crystalline phase verified
X-ray diffraction confirms target Li₂ZrCl₆ structure, free of significant secondary phases. Reference patterns: Li₃YCl₆ (JCPDS 44-0288) and LiCl (JCPDS 74-1972).
Performance Data:
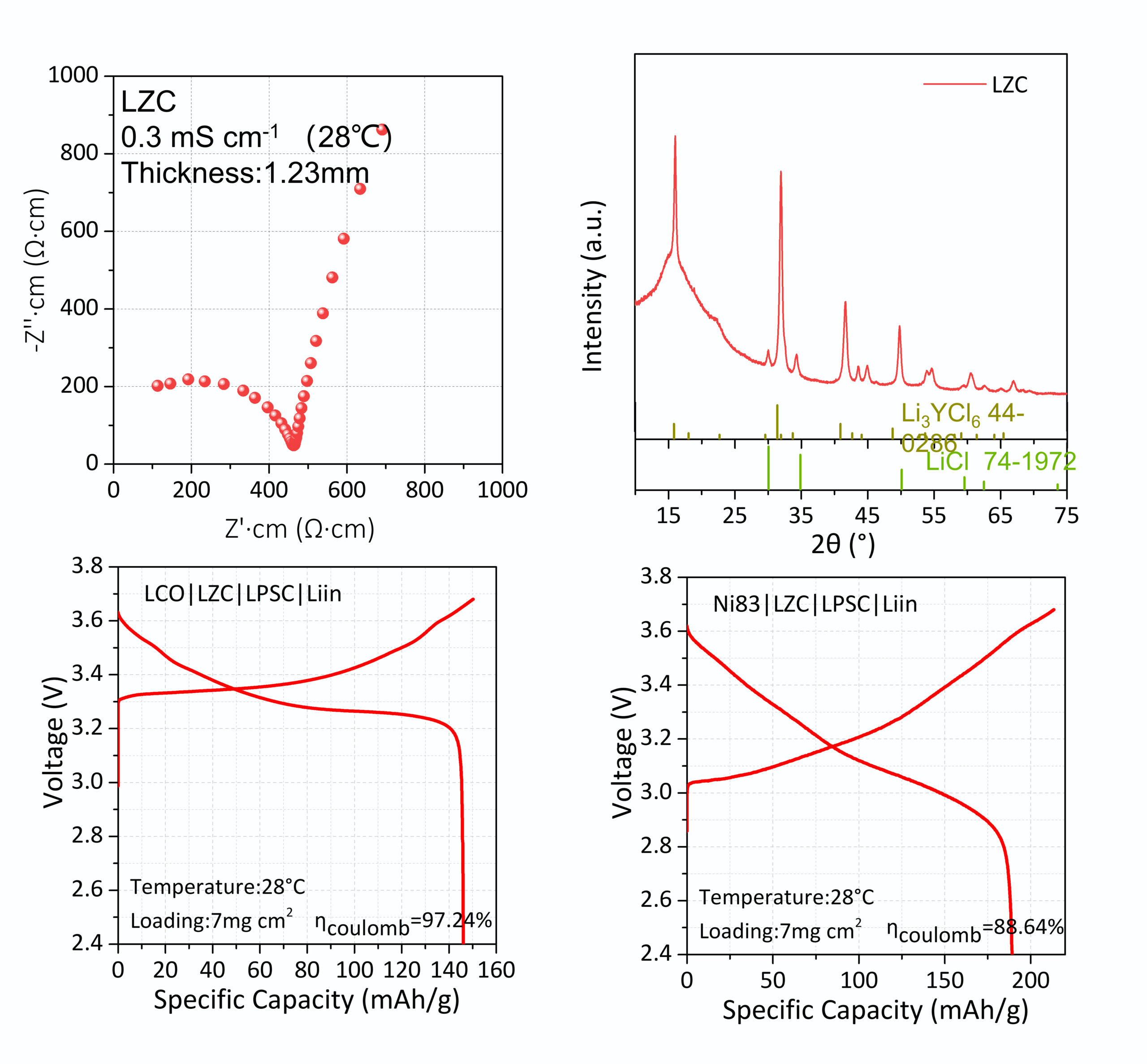
(Top left) EIS Nyquist plot of LZC pellet (1.23 mm) at 28 °C, showing 0.3 mS/cm ionic conductivity. (Top right) XRD pattern confirming Li₂ZrCl₆ phase. (Bottom left) LCO|LZC|LPSC|Li-In cell, ~150 mAh/g, 97.24% coulombic efficiency. (Bottom right) Ni83|LZC|LPSC|Li-In cell, ~190 mAh/g, 88.64% coulombic efficiency.
Packaging & Storage:
Vacuum-sealed under inert atmosphere in moisture-barrier aluminum-laminated bags. Store sealed in Ar or N₂ glovebox. LZC is moisture-sensitive — handle in dry environment with dew point < –40 °C. Reseal promptly after opening.
Safety:
For research and industrial use only. Wear PPE (gloves, masks, safety goggles). Refer to SDS for complete safety information.
Note: Values listed above are typical and for reference only. Performance may vary depending on cell architecture, cathode selection, electrolyte separator, and test protocol. For higher ionic conductivity, see our Al-Doped LZC Solid-State Electrolyte (0.6–0.8 mS/cm).




