SPAN Cathode Powder for Li–S Batteries | Global Supply
A polymer-bonded sulfur cathode in which sulfur is covalently linked into a pyrolyzed PAN backbone via C–S and N–S bonds — suppressing the polysulfide shuttle and enabling stable cycling in carbonate electrolytes.
Available from Xnergy Materials as a U.S.-based supplier with global shipping for academic and industrial research customers.


SPAN (sulfurized polyacrylonitrile) is a polymer-bonded sulfur cathode in which elemental sulfur is covalently linked into a pyrolyzed PAN backbone via C–S and N–S bonds. Unlike conventional sulfur or C/S composite cathodes, SPAN undergoes a quasi-solid-state conversion reaction that does not release soluble polysulfide intermediates — making it one of the very few sulfur cathode chemistries that operates stably in low-cost carbonate electrolytes. Xnergy's research-grade SPAN powder delivers > 50 wt% sulfur content and > 650 mAh/g at 0.1C, with standard 50 g / 100 g / 500 g packs available worldwide. SPAN's main trade-offs — lower accessible capacity than free sulfur, ~1.85 V average voltage, and 70–85% first-cycle CE — are discussed honestly in §02.
A Polymer-Bonded Sulfur Cathode for Li–S Research
SPAN — sulfurized polyacrylonitrile — is synthesized by thermally treating a mixture of elemental sulfur and polyacrylonitrile (PAN) at roughly 300–450 °C. During this pyrolysis, PAN dehydrogenates and partially cyclizes into a conjugated carbon–nitrogen heterocyclic backbone, while reactive sulfur species attack the polymer chain to form covalent C–S and N–S linkages. The resulting material is a black, electronically conductive solid in which the active sulfur is chemically bound rather than physically encapsulated.
"SPAN-based Li–S systems have been the subject of sustained and rapidly growing global research activity since the material was first reported by Wang et al. in 2002, with cumulative literature now exceeding 600 publications." — MDPI Batteries (2025), comprehensive review of SPAN cathodes
This structural feature distinguishes SPAN from the two other major sulfur cathode strategies:
- Elemental sulfur — highest theoretical capacity (~1672 mAh/g), but suffers from severe polysulfide dissolution, electronic insulation, and incompatibility with carbonate solvents.
- C/S composites (mesoporous carbon/sulfur, graphene/sulfur, hollow-carbon/sulfur, etc.) — host structures that physically confine sulfur. They improve cycling but cannot fully prevent polysulfide leakage over hundreds of cycles.
- SPAN — sulfur is part of the polymer; there is no free S₈ phase, no Li₂S₈/Li₂S₆/Li₂S₄ shuttle, and discharge proceeds through a topotactic, quasi-solid-state pathway.
The trade-off, which we discuss in §02, is that the covalent bonding limits accessible sulfur content and lowers the practical capacity ceiling relative to elemental-sulfur systems. Whether that trade-off is worthwhile depends on what the cell is being designed for — we lay out the cases below.
The Case for SPAN in Li–S R&D
SPAN occupies a distinctive position in Li–S research because it allows researchers to decouple two problems that are otherwise coupled: high sulfur utilization and electrolyte compatibility.
Carbonate-electrolyte compatibility
Conventional sulfur cathodes are restricted to ether electrolytes (DOL/DME with LiTFSI), because carbonate solvents (EC, DMC, EMC) react irreversibly with soluble polysulfides. SPAN's quasi-solid-state mechanism does not generate free polysulfides, so it cycles stably in standard Li-ion-type carbonate electrolytes. This is operationally significant: SPAN cells can be assembled on the same coating, drying, and electrolyte-filling lines used for NCM and LFP cells.
Suppressed shuttle and self-discharge
Because polysulfides are not solvated, SPAN cells exhibit very low self-discharge, and capacity fade is dominated by lithium-anode-side losses rather than cathode dissolution.
Mechanical and processing robustness
The conjugated polymer matrix gives the powder mechanical integrity and electronic conductivity, removing the need for the very high carbon loadings (often 30–40 wt%) typical of C/S composite electrodes.
What SPAN Is Not Good At
Any researcher considering SPAN should also weigh the well-documented limitations:
- Lower accessible capacity than free sulfur. Even with > 50 wt% sulfur, polymer-bonded SPAN typically delivers practical capacities around 650–700 mAh/g — well below the 1200–1500 mAh/g initial discharge sometimes seen with elemental S systems.
- Low average discharge voltage (~1.85 V). This caps cell-level energy density relative to NMC chemistries, even at high gravimetric capacity. Pairing strategy (Li-metal anode, lean electrolyte) matters considerably.
- First-cycle Coulombic efficiency is not unity. Initial irreversible capacity from PAN-side chemistry and SEI formation typically gives first-cycle CE in the 70–85% range. Pre-lithiation or sacrificial-additive strategies are common in the literature.
- Long-cycle failure is anode-driven. Pushing SPAN to 1000+ cycles is almost always a Li-metal-anode problem, not a cathode problem — but it remains a practical constraint when designing a full cell.
These constraints are intrinsic to SPAN chemistry, not specific to any supplier. We mention them so that experimental design starts from a realistic baseline.
When SPAN Is Not the Right Choice
To make the trade-off concrete, here are three research scenarios where another sulfur cathode chemistry is usually a better starting point:
- Maximizing sulfur utilization (> 80%). If your research target is approaching the 1672 mAh/g theoretical capacity of sulfur, polymer-bonded SPAN's accessible capacity ceiling is structurally limiting. Elemental S or high-S-loading C/S composites are more appropriate.
- Full-cell energy-density work requiring > 2.0 V average voltage. SPAN's ~1.85 V plateau caps achievable cell-level Wh/kg even at high gravimetric capacity. For practical pouch-cell energy-density benchmarking against NMC, this is a real constraint.
- Polysulfide-mechanism studies. If the explicit goal is to investigate polysulfide intermediates, redox mediators, or shuttle-suppressing additives, SPAN — which doesn't generate free polysulfides — is the wrong starting material by design.
In all three cases, the chemistry choice should follow the research question, not the other way around. We supply elemental sulfur and other cathode materials separately for these use cases; browse the full cathode catalog to see alternatives.
Three Standard Pack Sizes for R&D
Xnergy SPAN cathode powder is supplied in three standard pack sizes to support research at every stage — from initial coin-cell screening through pouch-cell scale-up. All packs share identical specifications and quality control; the only difference is quantity. Larger or custom batches are available on request.
SPAN · 50 g
- Standard specifications
- Argon-protected packaging
- 12-month shelf life (sealed)
- Ships within 1 week
SPAN · 100 g
- Standard specifications
- Argon-protected packaging
- 12-month shelf life (sealed)
- Ships within 1 week
SPAN · 500 g
- Standard specifications
- Argon-protected packaging
- 12-month shelf life (sealed)
- Ships within 1 week
All three packs are drawn from the same QC'd batches and ship under argon in hermetically sealed aluminum-foil pouches. For batches larger than 500 g, kilogram-scale pilot quantities, or technical inquiries about specific test protocols, please contact our team directly.
SPAN Across the Sulfur Cathode Landscape
Sulfur cathodes for Li–S research fall into three broad chemistry families. Each has its own performance trade-offs; SPAN occupies the polymer-bonded branch.
| Family | Examples | Practical Capacity | Carbonate Compatibility | Polysulfide Shuttle | Cycle Stability |
|---|---|---|---|---|---|
| Elemental sulfur | S₈ / Super P / PVDF | 1200–1500 mAh/g (initial) | ✗ | Severe | < 50 cycles typical |
| C/S composites | Mesoporous C/S, rGO/S, hollow-carbon/S | 800–1200 mAh/g (initial) | ✗ | Partial — leaks over cycles | 50–300 cycles |
| Polymer-bonded (SPAN) | SPAN and PAN-derived sulfur polymers | > 650 mAh/g (stable) | ✓ | Suppressed | 300–1500+ cycles |
Values represent typical ranges from peer-reviewed Li–S literature. Actual results depend strongly on cell architecture, electrolyte formulation, sulfur loading, and test protocol.
Representative Electrochemical Data
The figure below shows representative charge–discharge behavior measured on Xnergy SPAN in pouch-cell format. Two discharge formulations (SPAN 1 and SPAN 2) and the corresponding SPAN 1 charge curve are plotted across a specific-capacity range extending beyond 800 mAh/g.
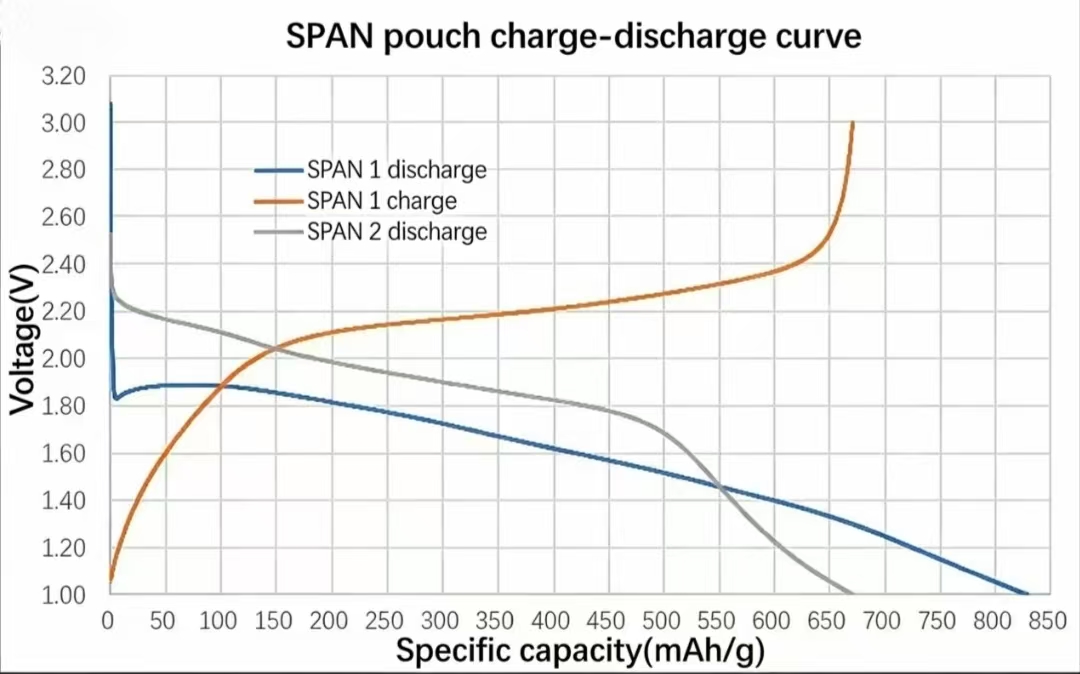
Key observations:
- SPAN 1 discharge — characteristic plateau at ~1.85–1.90 V in the early-capacity region, sloping gradually to ~0.85 V at ~820 mAh/g.
- SPAN 2 discharge — slightly elevated initial voltage (~2.25 V) with capacity extending to ~620 mAh/g.
- SPAN 1 charge — rises from ~1.05 V and reaches 3.00 V at ~660 mAh/g.
The single-plateau profile (rather than the two-plateau curve typical of elemental-S cells) is the electrochemical fingerprint of the quasi-solid-state conversion mechanism enabled by covalent C–S bonding. Per-batch electrochemical characterization reports are available with the technical data sheet on request.
Material Properties & Packaging
Values are typical and may vary by batch. A full Technical Data Sheet (TDS) with test methods and per-batch Certificate of Analysis is available on request.
Where SPAN Fits in Battery R&D
SPAN is intended primarily for advanced Li–S battery research at universities, national laboratories, and corporate R&D programs. Typical research applications include:
- Carbonate-electrolyte Li–S cells — using standard EC/DMC or EC/EMC formulations to study mechanisms inaccessible to elemental-S cells.
- High-loading pouch-cell prototypes — combining SPAN with lean-electrolyte protocols for energy-density studies.
- Quasi-solid-state mechanism studies — operando XRD, XAS, NMR, and Raman investigations of the C–S/N–S redox pathway.
- Lithium-metal anode pairing studies — SPAN's stable cathode side isolates anode-driven cycle-life losses for clean experimental attribution.
- Solid-state Li–S cells — pairing SPAN with sulfide or oxyhalide solid electrolytes (LNOC, LTOC).
- Coin-cell screening for new electrolyte additives — SPAN's reproducibility makes it a clean reference cathode for additive benchmarking.
For a complete Li–S coin cell, SPAN is typically combined with conductive carbon (Super P or Ketjen Black), a binder (PVDF or CMC/SBR), aluminum-foil current collector, polymer or glass-fiber separator, lithium metal foil anode, and a matched electrolyte. Xnergy supplies all of these components — see Related Materials below.
From the Field
Brief, anonymized reports from research customers using Xnergy SPAN. We do not publish institution names; the descriptions below are paraphrased with permission and intended only as directional reference for prospective users.
Both reports paraphrased with permission; institution-level confirmation is available on request, subject to confidentiality terms.
Beyond Standard Pack Sizes
For programs whose requirements exceed the 500 g standard pack — multi-batch R&D efforts, pouch-cell scale-up trials, or longer-term supply — Xnergy Materials offers bulk pricing, long-term supply agreements, and direct technical support. We work with research teams as a U.S.-based supplier and typically return quotes for bulk inquiries within one to two business days.
- Kilogram-scale pilot quantities (above 500 g) by quote
- Volume pricing for multi-batch and multi-quarter purchase commitments
- Per-batch characterization reports and Certificate of Analysis (CoA) on request
- Technical Data Sheet (TDS) and MSDS available with order confirmation
- Material Transfer Agreements (MTAs) for academic programs
- Long-term supply agreements for multi-year R&D programs
- Worldwide shipping with full export and hazmat documentation
Quote & Technical Inquiry
Tell us about your research program. We typically respond within one business day.
Frequently Asked Questions
SPAN is made by thermally reacting elemental sulfur with polyacrylonitrile at 300–450 °C, producing a polymer in which sulfur is covalently bonded into a carbon–nitrogen backbone via C–S and N–S linkages. Unlike pure sulfur or C/S composites — where sulfur is physically held in a host material — SPAN's chemistry produces no soluble polysulfide intermediates during cycling, which suppresses the shuttle effect and enables compatibility with carbonate electrolytes.
Conventional sulfur cathodes generate soluble lithium polysulfides during discharge. In carbonate solvents these polysulfides react irreversibly with the carbonate, destroying the cell within a handful of cycles. SPAN's covalent C–S/N–S bonding produces a quasi-solid-state pathway that never generates free polysulfides — so the carbonate-degradation reactions never occur.
Typical reversible capacity is > 650 mAh/g at 0.1C in a coin cell with a carbonate electrolyte. Cycle life depends heavily on electrolyte choice, sulfur loading, and depth-of-discharge protocol. Under standard test conditions Xnergy SPAN reaches ≥ 300 cycles at 80% retention; published literature reports 500–1500+ cycles with optimized electrolyte systems and additives such as LiNO₃ in ether-electrolyte cells.
First-cycle CE for SPAN typically falls in the 70–85% range depending on electrolyte and binder system. The irreversible capacity comes from PAN-backbone-side reactions and initial SEI formation. For full-cell energy-density work, pre-lithiation or sacrificial-additive strategies are commonly used and well documented in the literature.
The 50 g pack is a good fit for initial screening or one-time mechanism studies. The 100 g pack is the most common choice for ongoing R&D programs running multiple coin-cell campaigns or small pouch-cell prototyping. The 500 g pack is recommended for pouch-cell scale-up or multi-formulation comparison studies. All three packs share identical specifications and QC; only the quantity differs. For batches larger than 500 g or kilogram-scale pilot quantities, please contact us for a custom quote.
Standard SPAN starts at a 50 g minimum order. Available pack sizes are 50 g, 100 g, and 500 g. For batches larger than 500 g or kilogram-scale pilot quantities, please contact us for a custom quote.
Every batch is sealed under argon in hermetically closed aluminum-foil pouches. Standard pack sizes ship within one week to most destinations in the U.S., Europe, and Asia. We provide full export documentation and packaging compliant with IATA / ADR / DOT regulations. Sealed material has a 12-month shelf life at room temperature; after opening, transfer to a glovebox or dry room (dew point < –40 °C).
SPAN itself is more tolerant of brief ambient exposure than reactive materials such as lithium metal or sulfide solid electrolytes. However, prolonged humidity exposure introduces moisture that degrades electrochemical performance. After opening, store and handle in inert atmosphere.
A typical Li–S coin cell stack includes SPAN cathode powder + conductive carbon (Super P or KB) + binder (PVDF or CMC/SBR) + aluminum-foil current collector; a polymer or glass-fiber separator; lithium-metal foil anode; and a matched electrolyte (carbonate or ether). Xnergy supplies all of these components — see Related Materials.
SPAN is not ideal when the research goal is to maximize sulfur utilization above ~80%, when an average operating voltage above 2.0 V is required for full-cell energy-density targets, or when the study explicitly aims to investigate polysulfide-mediated mechanisms (intermediates, redox mediators, shuttle suppressors). In those cases, elemental sulfur or C/S composite cathodes are more appropriate starting materials. See §02 — When SPAN Is Not the Right Choice above for details.
Out of respect for our customers' research confidentiality, we do not publish customer lists. References from prior research customers are available on a case-by-case basis under appropriate confidentiality terms. Where peer-reviewed publications have used Xnergy SPAN, citations can be provided on request.
Other Materials from Xnergy
Researchers often pair SPAN with other components to assemble a complete Li–S or solid-state Li–S cell. The following materials are also stocked at Xnergy:
Battery-Grade Electrolytes
LiPF₆, LiFSI, LiTFSI custom formulations; carbonate and ether systems for Li–S and Li-ion research.
High-Purity Lithium Metal
Lithium foil, strip, and alloys — argon-packaged for safe delivery and glovebox handling.
Coin Cells & Components
CR2016, CR2025, CR2032 hardware for electrode screening and cycling studies.
Electrode Binders
PVDF, CMC, SBR, and specialty binders optimized for slurry casting and high-loading electrodes.
LNOC, LTOC & Beyond
Niobium and tantalum oxychloride solid electrolytes for solid-state Li–S research.
Dry Pouch Cells
Pre-fabricated pouch-cell formats for scale-up from coin to pouch prototyping.
Selected Literature on SPAN
For researchers new to SPAN chemistry, the following references provide background on synthesis, ion-transport mechanisms, and cell-level performance:
- Wang, J.; Yang, J.; Xie, J.; Xu, N. (2002). A novel conductive polymer–sulfur composite cathode material for rechargeable lithium batteries. Advanced Materials, 14(13–14), 963–965 — first report of the SPAN cathode concept.
- Ali, A. et al. (2025). Sulfurized Polyacrylonitrile for Rechargeable Batteries: A Comprehensive Review. MDPI Batteries, 11(8), 290 — covers 600+ publications, 2002–2025.
- Lee, J. et al. (2024). Realizing High-Energy and Long-Life Li/SPAN Batteries. Joule — practical cell-level energy density analysis.
- Li, S. et al. (2019). Ether-Compatible SPAN Cathode via Selenium Doping. Nature Communications, 10, 725 — kinetics enhancement via Se substitution.
- Lithium–Sulfur Battery — Wikipedia overview of Li–S electrochemistry, history, and remaining challenges.
References listed for general scientific background. Per-batch characterization reports specific to Xnergy SPAN material lots are available with the technical data sheet on request.
