Background
With the global adoption of electric vehicles in hot regions and the rapid expansion of ultra-fast charging infrastructure, lithium-ion batteries are facing increasingly severe thermal challenges. Driven by both policy support and market demand, researchers and industry are pursuing higher energy density and faster charging. As a result, the operating temperature of batteries continues to rise.
At present, conventional commercial battery systems show a clear decline in stability when the temperature exceeds 60°C. Under high-temperature conditions, battery electrolytes tend to decompose and volatilize. This not only accelerates capacity decay, but may also trigger thermal runaway.
From onboard power batteries in hot climates to outdoor energy storage systems, the high-temperature tolerance of electrolyte materials has become a key bottleneck for battery safety and performance. Therefore, designing high-temperature electrolytes that can work stably at 80°C or even higher has become a core challenge for industry. This is also critical for expanding the thermal safety boundary of battery systems and enabling reliable battery operation in all-weather and all-region applications.
Work Overview
Recently, this team reported a strategy that regulates the solvation structure by utilizing the different temperature responses of various lithium salts. Based on this strategy, they designed a low-cost temperature-adaptive electrolyte, named TAE, which enables liquid lithium-ion batteries to operate normally at elevated temperatures.
Without changing the solvent system, TAE takes advantage of the interactions between Li⁺ and different anions at different temperatures. In this way, the solvation structure of the electrolyte can dynamically adapt to changes in the external environment. It can also form inorganic-rich interphases under different temperature conditions.
As a result, lithium-ion batteries using TAE show excellent electrochemical performance over a wide temperature range. At room temperature, TAE forms an F-rich inorganic interphase. At elevated temperature, it forms a B-rich inorganic interphase.
With this electrolyte, lithium-ion batteries achieved a capacity retention of 89.6% after 500 cycles at 4.5 V and room temperature. At 80°C, the capacity retention remained as high as 90.8% after 450 cycles. In addition, pouch cells using the TAE electrolyte delivered 86.1% capacity retention after 100 cycles at 100°C.
This work was published in the Journal of the American Chemical Society under the title Temperature-Adaptive Electrolyte Enables Stable Cycling of Liquid Lithium Pouch Cells at ≥100 °C.
Figure 1. Schematic illustration of the concept of a temperature-adaptive electrolyte
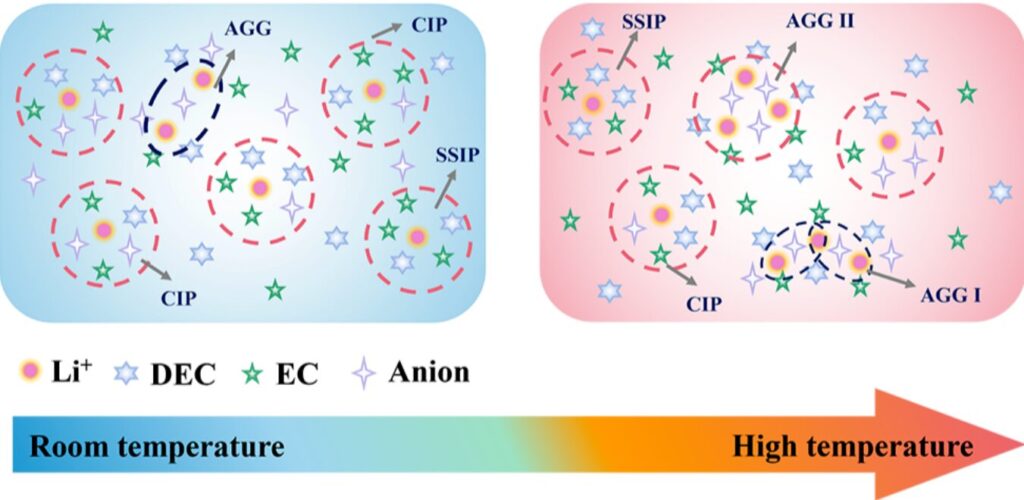
Figure 2. Design principles of the temperature-adaptive electrolyte
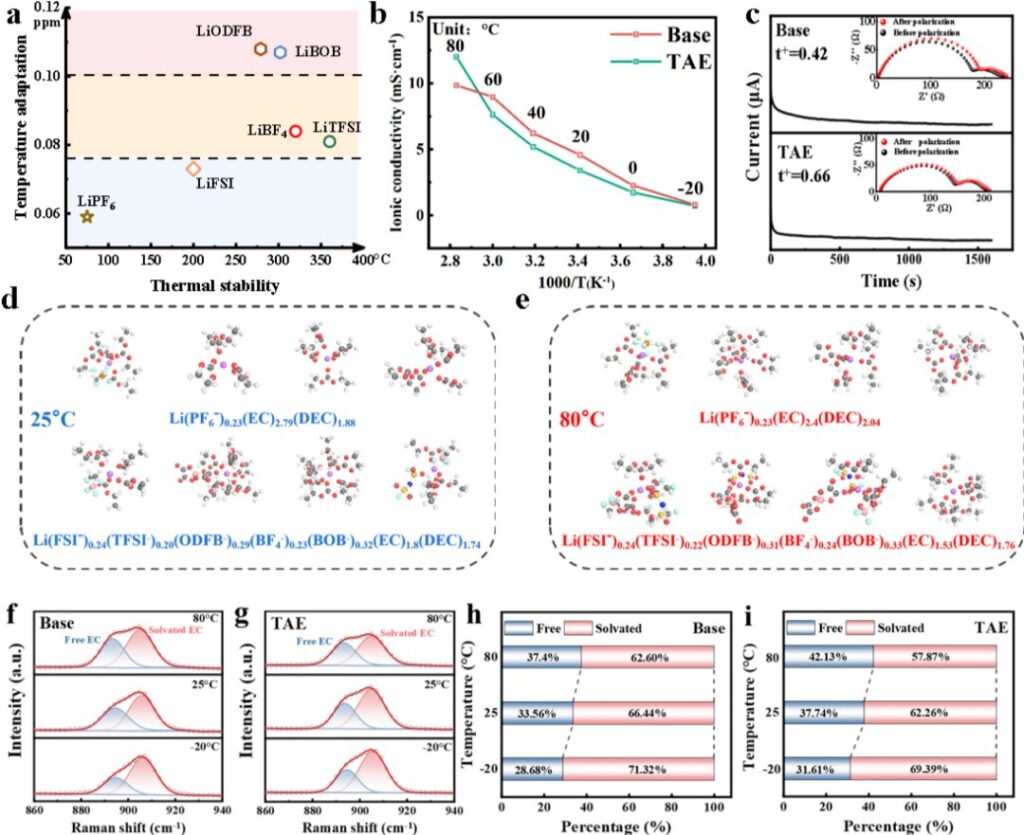
The team first systematically studied the thermal stability and temperature sensitivity of six lithium salts. Based on their behaviors, the six salts were classified into three major categories. This classification served as the basis for designing the TAE electrolyte.
At low temperature and room temperature, the ionic conductivity of TAE was comparable to that of the base electrolyte, namely 1M LiPF₆ in EC/DMC/DEC=1:1:1 (v/v/v) Electrolyte. At elevated temperatures, however, the ionic conductivity of TAE was clearly higher than that of the base electrolyte.
MD simulations and variable-temperature Raman results further revealed that the proportions of LiODFB and LiBOB in the solvation structure of TAE increased with rising temperature. This result agrees well with the original design concept, which proposed that these lithium salts should show stronger temperature responsiveness. These results confirm that TAE is indeed a temperature-adaptive electrolyte.
Figure 3. Analysis of the solvation structure of the TAE electrolyte
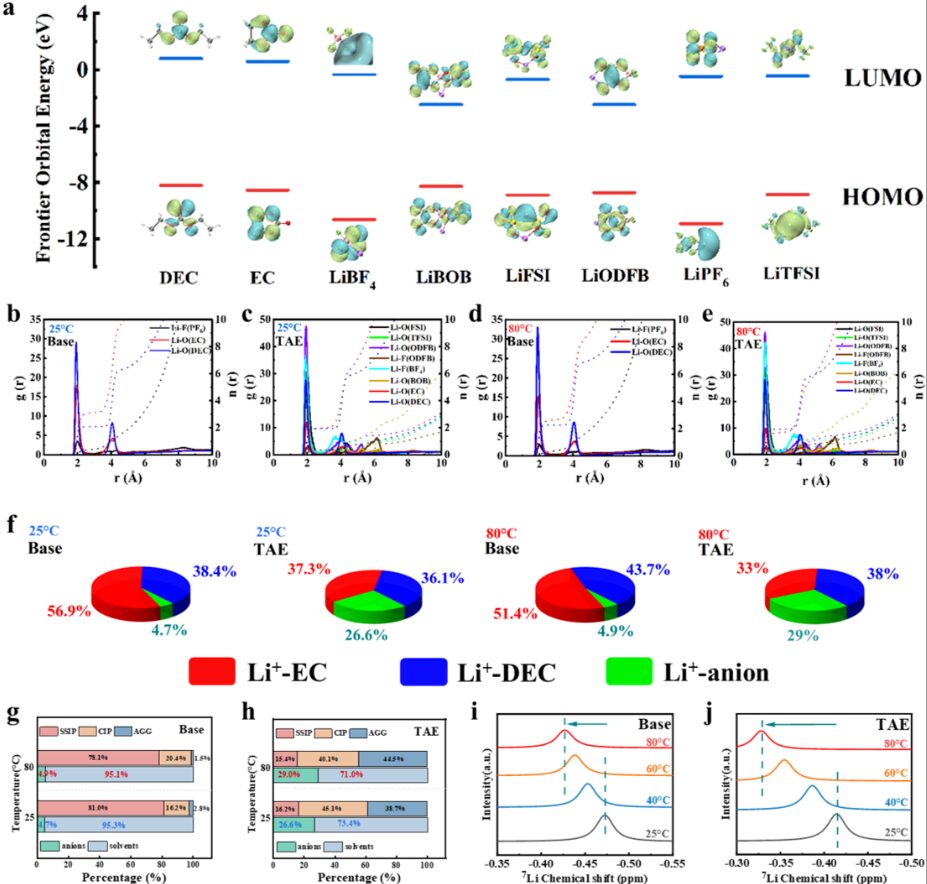
The team further analyzed the solvation structure of TAE by using MD simulations and variable-temperature NMR. The results showed that, at both room temperature and elevated temperature, the proportion of anions in the solvation structure of TAE was much higher than that in the base electrolyte. This indicates that TAE has a weakly solvated structure.
More importantly, as the temperature increased, the proportion of anions in TAE rose from 26.6% to 29%. In contrast, the anion proportion in the base electrolyte remained almost unchanged. This difference suggests that the solvation structure of TAE can automatically respond to changes in the external environment.
Figure 4. Electrochemical performance analysis of the base electrolyte and TAE electrolyte at room temperature
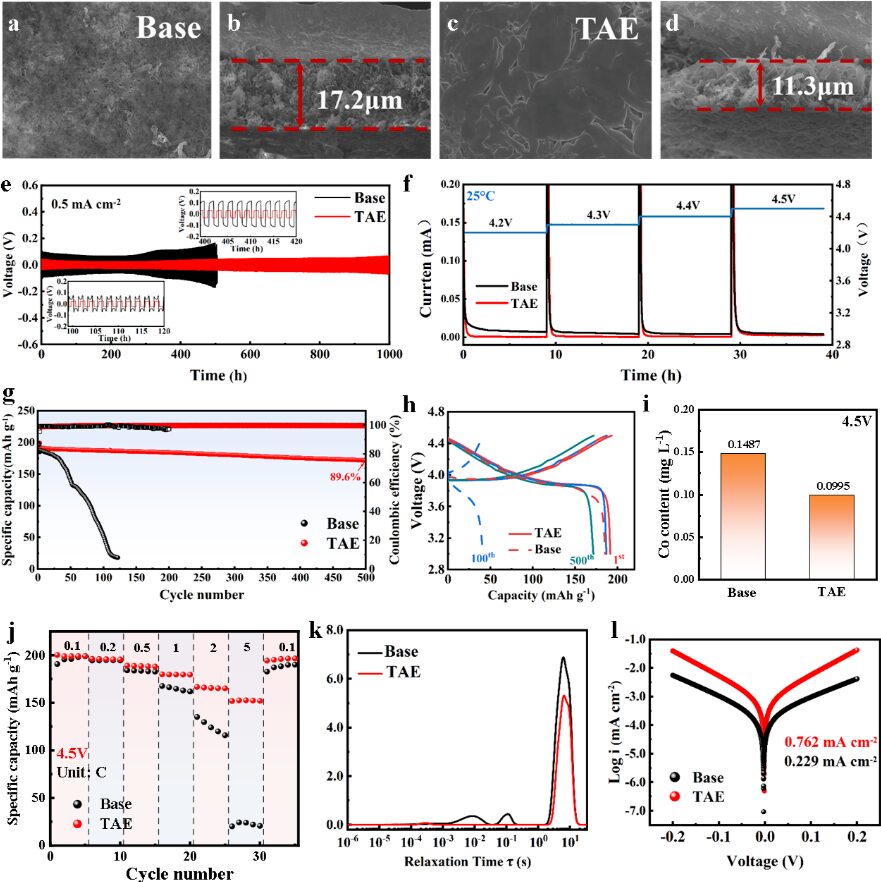
The team assembled Li||Cu cells and Li symmetric cells using both the base electrolyte and the TAE electrolyte. The results demonstrated that TAE has good compatibility with the lithium metal anode.
Compared with the base electrolyte, LCO||Li coin cells using TAE showed significantly reduced transition-metal ion dissolution under high-voltage operation at 4.5 V and room temperature. These cells maintained 89.6% of their capacity after 500 cycles. They also exhibited excellent electrochemical and kinetic performance.
For related LCO cathode sheet materials, Xnergy also provides products for lithium-ion battery research.
Figure 5. Electrochemical performance analysis of the base electrolyte and TAE electrolyte at elevated temperatures
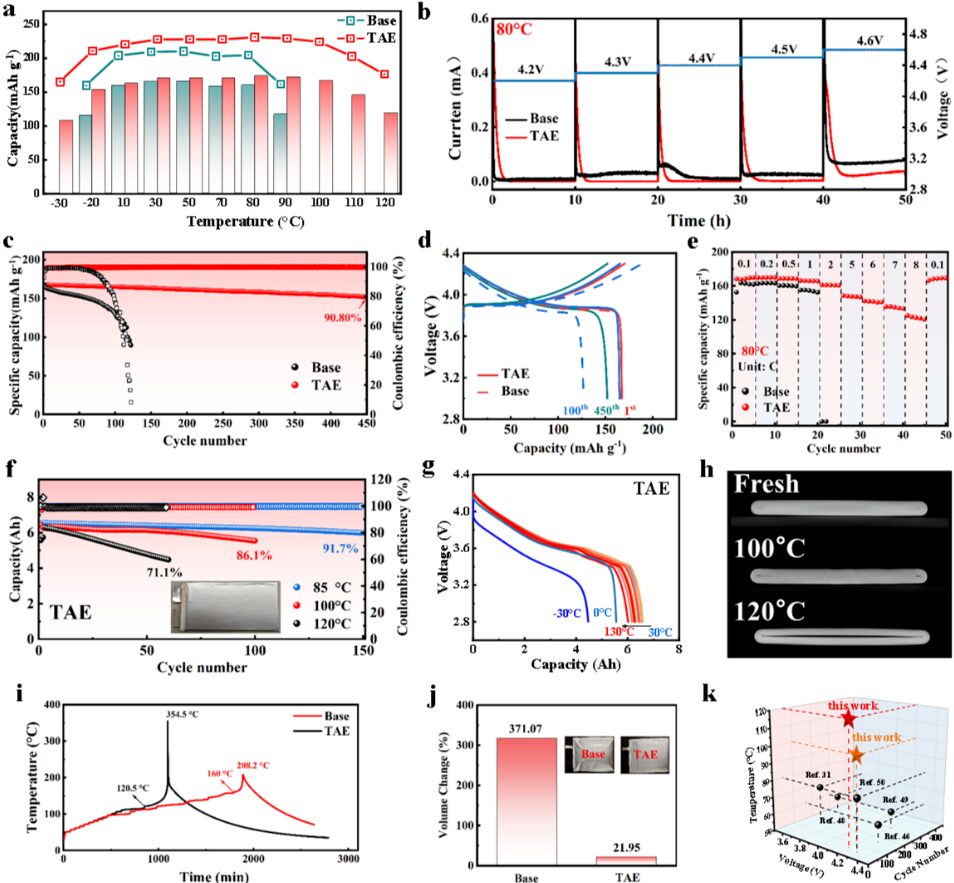
The team systematically evaluated the high-temperature performance of TAE. At 80°C, LCO||Li coin cells using the TAE electrolyte achieved a capacity retention of 90.8% after 450 cycles.
Furthermore, pouch cells filled with the TAE electrolyte cycled stably for 100 cycles at 100°C and maintained 86.1% of their capacity. These results demonstrate the excellent high-temperature cycling stability of the TAE electrolyte.
The team also evaluated the safety performance of TAE-filled pouch cells through ARC, CT, and related tests. Compared with the base electrolyte, TAE significantly increased the thermal runaway temperature. It also clearly reduced the swelling rate during high-temperature cycling.
These results indicate that TAE improves the safety of lithium-ion batteries under elevated-temperature conditions. They also highlight the practical application potential of this electrolyte strategy.
For related lithium metal materials, Xnergy offers battery-grade lithium products for advanced battery research.
Figure 6. Interfacial analysis of the TAE electrolyte at different temperatures
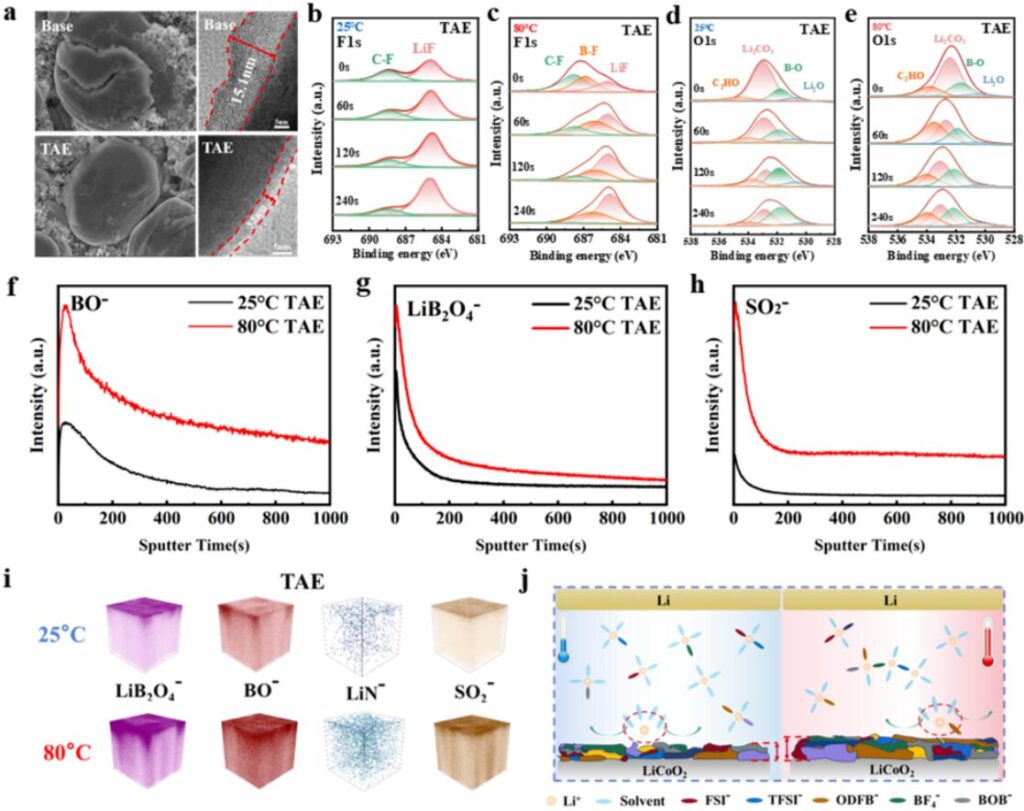
The team analyzed the interfacial compositions of cells assembled with the TAE electrolyte after cycling at different temperatures. At 25°C, cells using TAE tended to form an inorganic interphase rich in F. At 80°C, they tended to form an inorganic interphase rich in B.
This result is consistent with the original design concept. It further confirms that the solvation structure of the TAE electrolyte can change with the external environment. In this way, the electrolyte can dynamically form the most suitable and stable interphase for different conditions.
Core Conclusion
This work takes advantage of the different temperature responses of various lithium salts and uses them to regulate the interactions between Li⁺ and anions at different temperatures. Based on this concept, the team designed a low-cost temperature-adaptive electrolyte that can adapt to external environments.
Without changing the solvent system, the solvation structure of this electrolyte evolves with temperature. As a result, it can dynamically form the most suitable interphase under different conditions. Therefore, it not only enables stable high-voltage cycling at room temperature, but also delivers excellent cycling performance under extreme high-temperature conditions.
For related battery molds and liquid battery molds, please visit Xnergy Materials.
