Background and Challenges
Lithium metal batteries (LMBs), owing to their ultrahigh theoretical energy density, are widely recognized as one of the most promising next-generation energy storage technologies for advanced lithium battery research, high-energy-density batteries, and practical energy storage systems. However, their real-world application is still severely restricted by interfacial instability and safety concerns. Under harsh operating conditions, particularly at high voltages (≥4.3 V) and during prolonged cycling, the lithium metal anode is highly susceptible to dendrite growth and internal short circuits. Meanwhile, high-nickel cathodes such as NCM811 cathode materials suffer from transition-metal dissolution and lattice collapse, leading to rapid capacity fading and poor long-term stability.
Gel polymer electrolytes (GPEs), which combine the high ionic conductivity of liquid electrolytes with the mechanical robustness of polymer matrices, have emerged as promising candidates for next-generation lithium battery electrolyte development. In particular, in situ polymerization enables intimate interfacial contact between the electrolyte and electrodes, which is highly desirable for solid-state battery research, lithium metal battery interface engineering, and high-voltage battery applications. Nevertheless, the solvation structures of current GPEs remain insufficiently understood, making it difficult to simultaneously optimize ion-transport efficiency and interfacial stability. Conventional strategies have mainly focused on solvent engineering, such as introducing fluorinated solvents, while overlooking the critical role of the polymer framework in regulating Li-ion solvation and transport. As a result, existing GPEs still suffer from limited cycle life and insufficient energy density under high-loading and high-voltage conditions.
Literature Context
Previous studies have improved the stability of LMBs by optimizing electrolyte compositions or constructing artificial interfacial layers. However, most of these advances have only been demonstrated in low-loading coin cells, making them difficult to translate into practical high-capacity pouch cells. For example, conventional liquid electrolytes exhibit high ionic conductivity, but suffer from leakage and poor dendrite suppression. Non-fluorinated GPEs can improve interfacial contact, yet strong coordination between the polymer and Li ions often hinders ion transport, limiting their suitability for high-performance lithium metal batteries and high-voltage NCM811 cells.
In this work, a polymer-framework-mediated “pseudo-diluent” strategy is proposed. By introducing a trifluoromethyl (-CF₃)-containing fluorinated polymer (TF) into a highly concentrated ether electrolyte (HCE) through in situ polymerization, a fluorinated gel electrolyte, denoted as TF+HCE gel polymer electrolyte, is constructed. The -CF₃ groups regulate the Li-ion solvation structure in a non-coordinating manner, thereby lowering the desolvation energy barrier while promoting the formation of an inorganic-rich solid electrolyte interphase (SEI) and cathode electrolyte interphase (CEI). Such a design is highly relevant to the development of fluorinated gel electrolytes, high-voltage lithium battery electrolytes, and advanced electrolyte materials for lithium metal batteries.
Under lean-lithium conditions (45 μm), high areal capacity (2.09 mAh cm⁻²), and high voltage (4.4 V), this strategy enables highly stable cycling. Coin cells achieve coulombic efficiencies (CEs) of ≥98.73% over 1000 cycles, while a NCM811 / lithium metal pouch battery dry cell demonstrates a promising pathway toward practical high-energy-density lithium metal pouch cells.
Key Innovations
1. Conceptual Innovation
A fluorinated polymer framework is proposed for the first time as a “pseudo-diluent” in a gel polymer electrolyte for lithium metal batteries, where non-coordinating -CF₃ groups modulate the solvation structure and simultaneously address the trade-off between ion transport and interfacial stability.
2. Interfacial-Regulation Innovation
The TF+HCE electrolyte induces the formation of a LiF-rich SEI on the anode and a uniform CEI layer on the cathode, enabling dual interfacial protection that suppresses dendrite growth and cathode dissolution without sacrificing ionic conductivity. This feature is particularly valuable for lithium metal materials, high-voltage cathode systems, and advanced battery electrolyte platforms.
3. Performance Breakthrough
The electrolyte delivers a room-temperature ionic conductivity of 4.02 mS cm⁻¹, a Li-ion transference number of 0.63, and an electrochemical stability window of 5.3 V, demonstrating strong potential for high-voltage lithium metal batteries, NCM811 full cells, and advanced battery electrolyte materials.
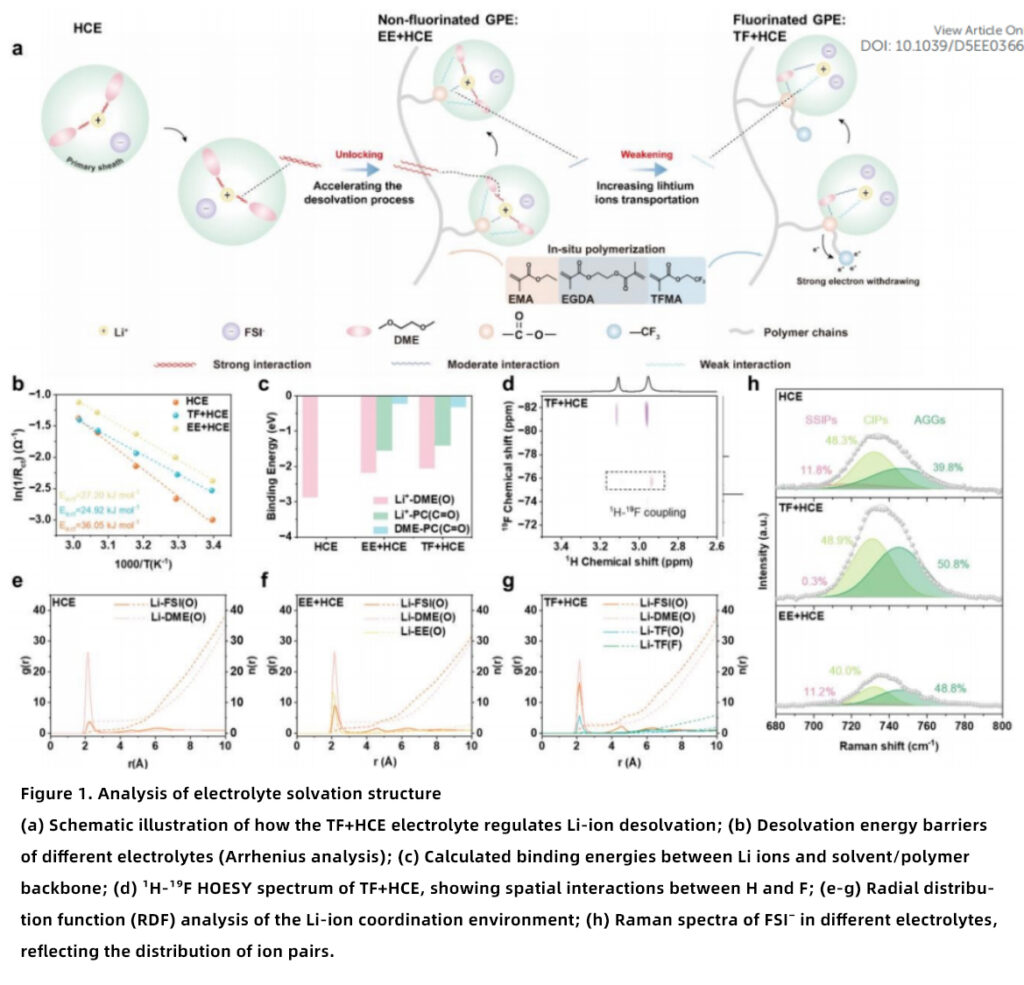
Mechanistic Insights into Solvation Regulation
This work systematically clarifies how the TF framework regulates the solvation structure in a fluorinated polymer electrolyte system.
Reduced Desolvation Energy Barrier
The desolvation energy of TF+HCE (24.92 kJ mol⁻¹) is significantly lower than that of HCE (36.05 kJ mol⁻¹) and EE+HCE (27.20 kJ mol⁻¹). The electron-withdrawing -CF₃ groups weaken the coordination between Li ions and ester groups, reducing desolvation resistance and improving Li-ion transport kinetics.
Optimized Ion Coordination Environment
RDF analysis shows that in TF+HCE, the coordination number between Li ions and DME decreases to 3.12, while that between Li ions and FSI⁻ increases to 2.58, forming a weakly solvated, anion-enriched electrolyte structure, which is highly beneficial for stable SEI/CEI formation in lithium metal batteries.
Regulated Ion-Pair Distribution
Raman spectroscopy reveals that TF+HCE exhibits the highest proportion of contact ion pairs (CIPs) and aggregates (AGGs), promoting anion participation in SEI formation. This behavior is highly advantageous for LiF-rich interphase engineering and stable lithium metal anodes.
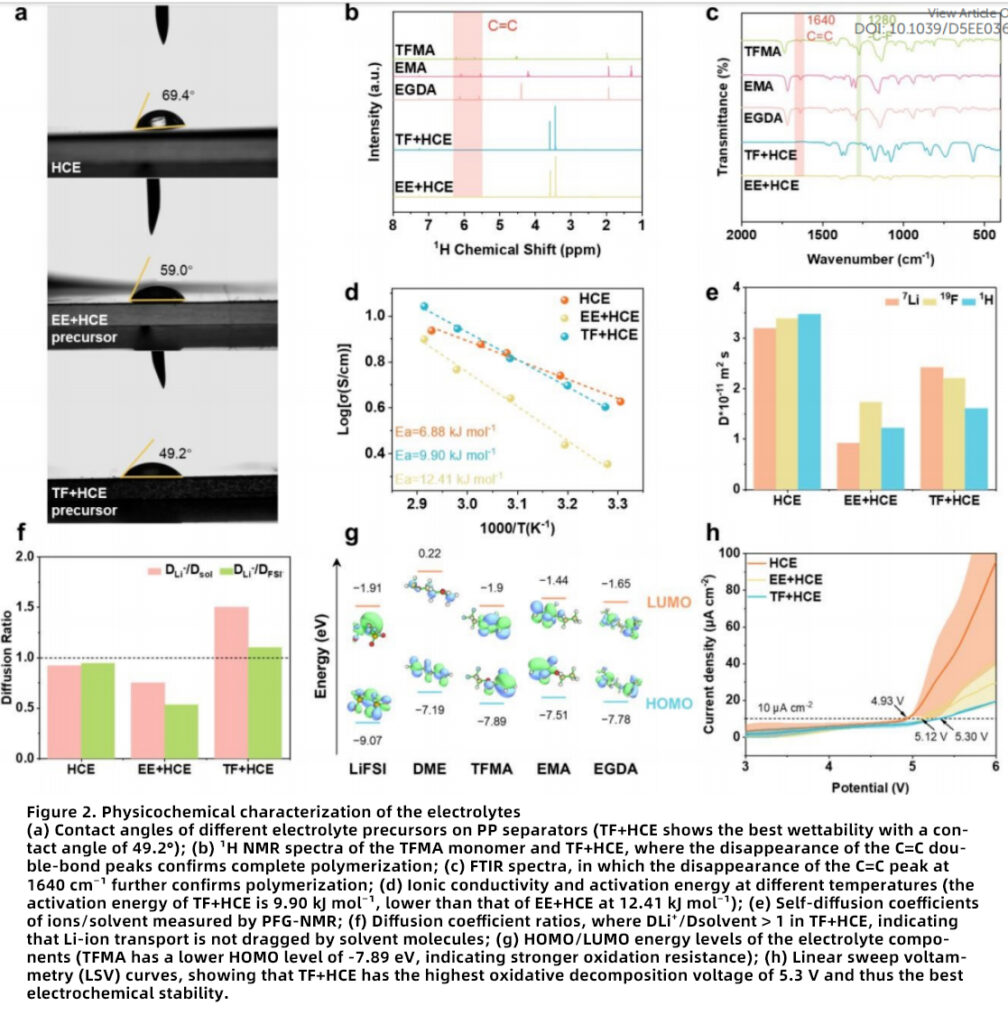
Physicochemical Advantages of TF+HCE
The physicochemical superiority of TF+HCE further highlights its value as a high-performance gel polymer electrolyte.
Wettability and Polymerizability
The TF+HCE precursor shows the smallest contact angle (49.2°), outperforming HCE and EE+HCE, and can be fully polymerized within 24 h at 60°C, indicating strong compatibility with existing battery manufacturing processes and in situ polymer electrolyte fabrication.
Ion-Transport Capability
At room temperature, TF+HCE achieves an ionic conductivity of 4.02 mS cm⁻¹, close to that of HCE, while delivering a higher Li-ion transference number of 0.63. This makes it attractive for fast Li-ion transport, high-rate lithium batteries, and long-cycle electrolyte systems.
Thermal Stability and Safety
TGA and flame testing confirm the superior thermal stability and flame-retardant behavior of TF+HCE, making it a safer candidate for high-safety lithium battery electrolytes and advanced energy storage systems.
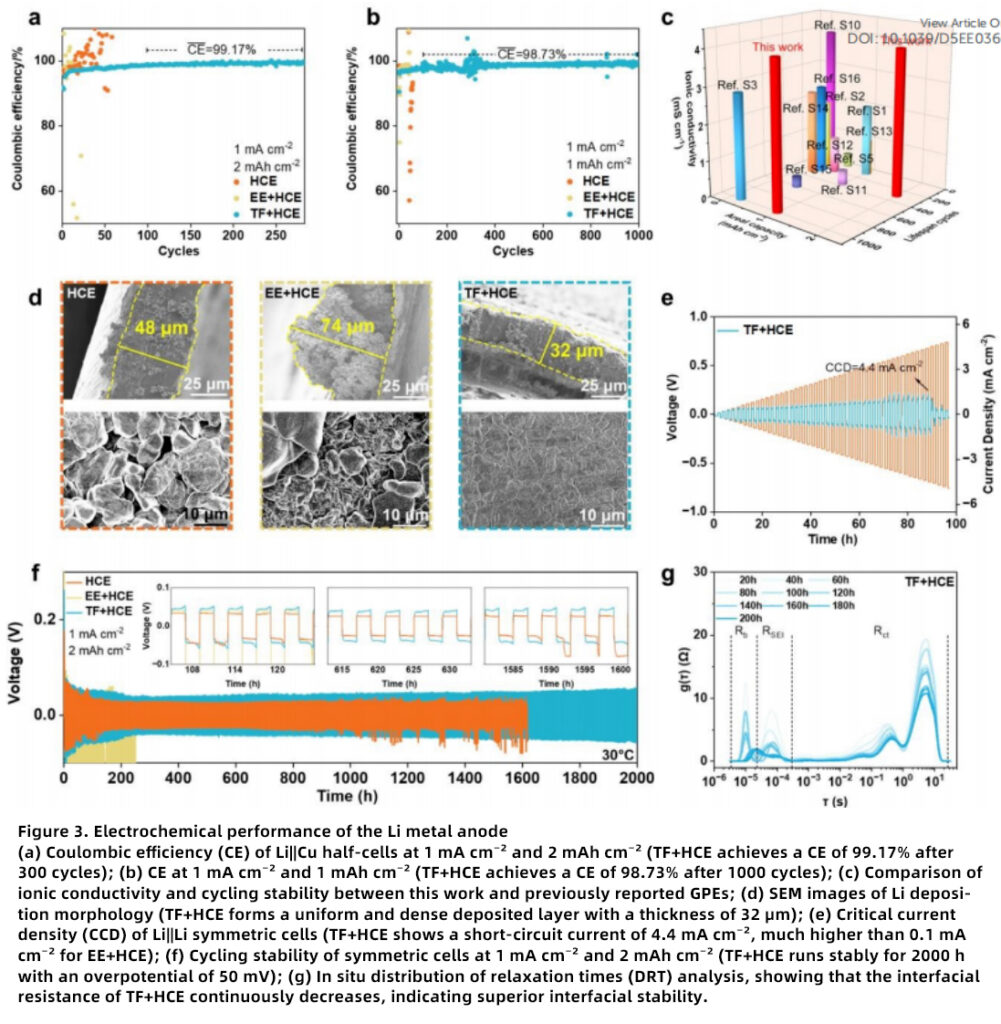
Protection of the Lithium Metal Anode
The TF+HCE electrolyte exhibits exceptional capability in stabilizing the lithium metal anode.
High Reversibility
In Li||Cu half-cells, TF+HCE maintains stable CE over 1000 cycles, highlighting its effectiveness as an electrolyte for lithium metal anode protection.
Uniform Lithium Deposition
SEM observations confirm dense and uniform lithium deposition in TF+HCE, demonstrating its suitability for dendrite-free lithium deposition and stable lithium plating/stripping.
Long-Term Cycling Stability
Li||Li symmetric cells cycle stably for 2000 h, underscoring the long-term durability of this advanced lithium metal battery electrolyte.
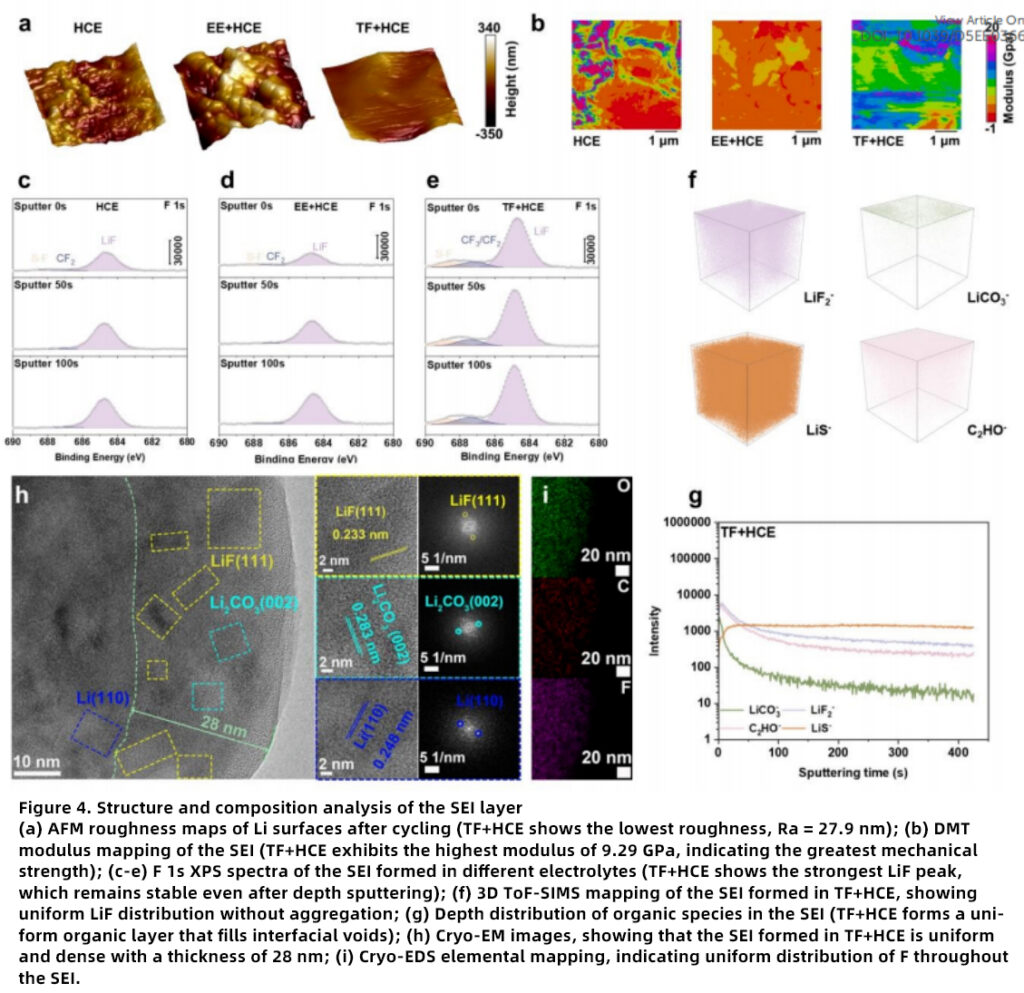
Superior SEI Properties
The SEI formed in TF+HCE exhibits several desirable characteristics for lithium metal battery interface stabilization.
High Mechanical Strength
The SEI modulus reaches 9.29 GPa, indicating excellent capability to resist dendrite-induced stress.
Stable Composition
XPS confirms the formation of a LiF-rich SEI layer, which is widely recognized as a key feature of high-performance fluorinated electrolytes.
Uniform Structure
Cryo-EM observations reveal a continuous and dense interphase, favorable for uniform ion flux distribution and long-life lithium metal batteries.
Practical Value in Full Cells
The practical relevance of TF+HCE is demonstrated in Li||NCM811 full-cell research and pouch-cell battery systems.
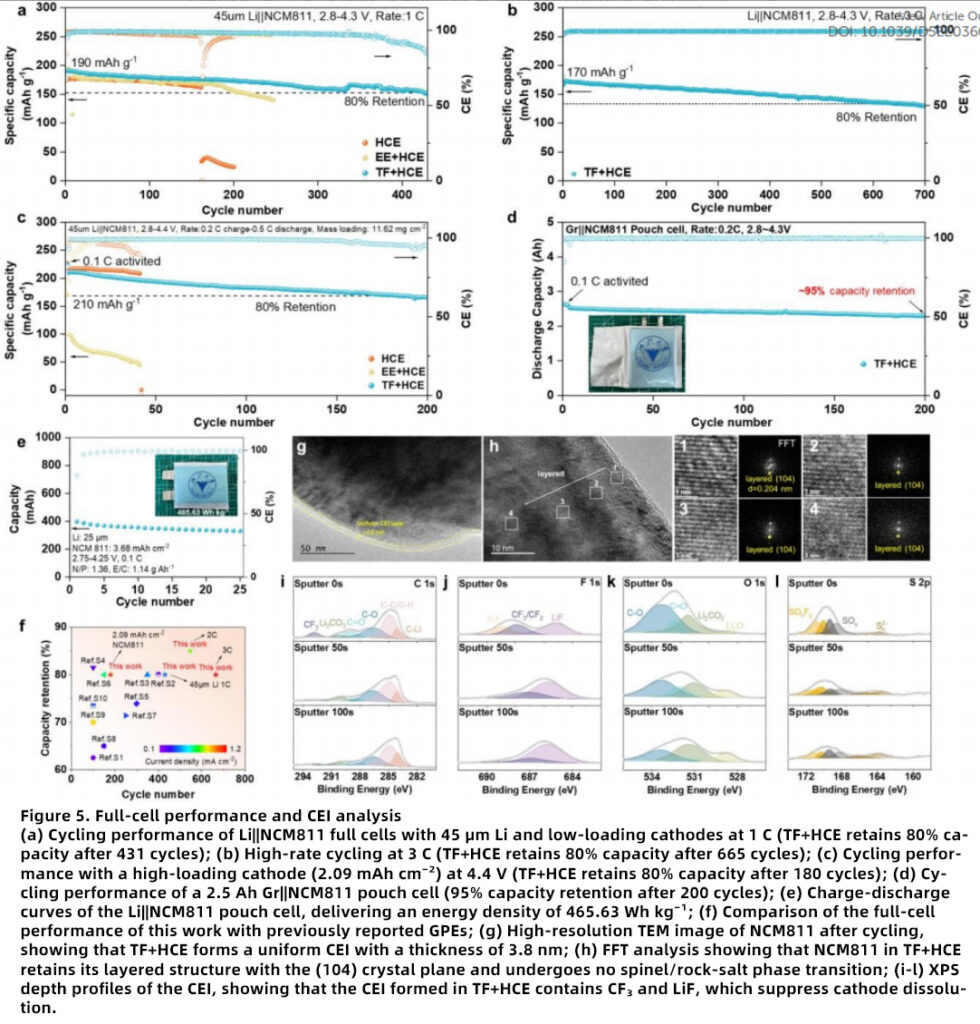
High-Voltage Stability
At 4.4 V, Li||NCM811 full cells with TF+HCE retain 80% capacity after 180 cycles, showing strong compatibility with high-voltage cathode materials and NCM811 battery systems.
High-Rate Capability and Long Lifespan
At 3 C, TF+HCE retains 80% capacity after 665 cycles, outperforming control electrolytes and highlighting its value for high-rate lithium batteries and long-cycle battery electrolytes.
Pouch-Cell Performance
A 2.5 Ah Gr||NCM811 pouch cell retains 95% capacity after 200 cycles with an average CE of 99.6%, demonstrating strong promise for practical pouch-cell applications and high-energy-density lithium battery commercialization.
Cathode Protection
TF+HCE forms a uniform CEI on NCM811, suppressing structural degradation and transition-metal dissolution, which is highly beneficial for high-nickel cathode protection and advanced cathode-electrolyte interface engineering.
In addition, its compatibility with solid-state electrolyte membrane, oxide solid electrolyte powder, and solid-state electrolytes for lithium battery research further demonstrates its value across both liquid and solid-state battery platforms.
For cell assembly and interfacial evaluation, solid-state battery molds and ceramic solid-state battery assembly mold can also support reproducible laboratory testing and advanced cell-development workflows.
Conclusion
This work presents a fluorinated gel electrolyte containing -CF₃ groups, denoted as TF+HCE, and proposes a new pseudo-diluent strategy for lithium metal battery electrolyte design. By weakening the strong interactions between Li ions and solvents/polymers in a non-coordinating manner, the TF framework reduces the desolvation energy barrier and promotes the formation of a LiF-rich SEI and a uniform CEI, thereby simultaneously improving interfacial stability and ion-transport efficiency.
As a result, the electrolyte delivers outstanding overall performance, including a room-temperature ionic conductivity of 4.02 mS cm⁻¹, a Li-ion transference number of 0.63, and an electrochemical stability window of 5.3 V. Li||Cu half-cells maintain CE ≥ 98.73% over 1000 cycles, Li||NCM811 full cells retain 80% capacity after 665 cycles at 3 C, and pouch cells achieve an energy density of 465.63 Wh kg⁻¹ with 95% capacity retention after 200 cycles.
These results establish TF+HCE as a highly promising fluorinated gel polymer electrolyte for high-energy-density lithium metal batteries, lithium metal materials platform, NCM811 battery research solution, and dry pouch cell development route for next-generation battery research and commercialization.

